A research group that includes Professor Makoto Tsuda and graduate student (at the time of the research) Ryoichi Tajima, graduate student (at the time of the research) Keisuke Koga and graduate student (at the time of the research) Yu Yoshikawa of the Department of Molecular and System Pharmacology, Graduate School of Pharmaceutical Sciences, Kyushu University; and Professor Toshiharu Yasaka of the Department of Health and Nutrition, Faculty of Health Sciences, Niigata University of Health and Welfare, discovered in an experiment using rats that activity by NPY nerve cells that are localized on surface of the spinal cord declines following neurological damage, and revealed for the first time in the world that this is the cause of neuropathic allodynia, which is difficult to relieve with morphine. Tsuda says, "We want to find an existing medicine that boosts nerve activity from the library, and move ahead with a physician-led clinical trial." The research results were published in PNAS Online.
Kyushu University and others shed light on allodynia, when "even touching is painful"
In many living things harmful stimuli are accurately transmitted as pain signals and stimuli such as touch are accurately transmitted as touch signals. However, in some cases chronic pain (neuropathic pain) that cannot be brought under control with mainstream medicines such as anti-inflammatories, antipyretics and analgesics appears as a result of cancer, diabetes, shingles, strokes, postoperative sequelae, and sequelae arising from spinal cord and other injuries. The cause of this has not been specified. In particular, allodynia, when even a light touch of the skin produces pain, is a troublesome condition for which even morphine is ineffective.
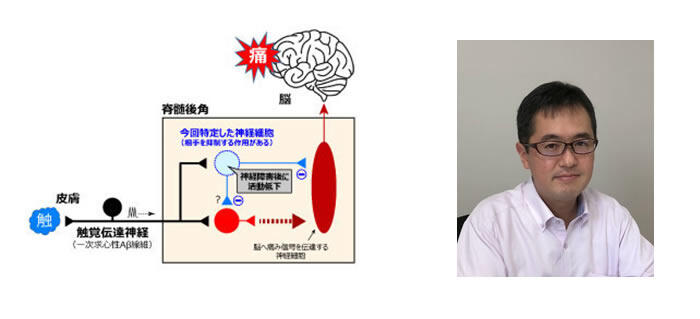
In the nerve cells of the spinal cord dorsal horn of rats, there are 15 varieties of excitatory neuron and 15 varieties of inhibitory neuron. Of these, the research group focused on the nerves that express neuropeptide Y (NPY). Using an optogenetic method, the group built a rat experimental system capable of controlling the NPY promoter found within the spinal cord dorsal horn.
Ordinarily, sharp pain such as being pricked with a needle reaches the spinal cord from the skin's high-threshold mechanoreceptors. From there the pain is conveyed to the brain through the lateral spinothalamic tract and other pathways. NPY nerves have the ability to strongly inhibit the nerves that convey pain to the brain, and so the research team believed that allodynia may appear when NPY nerve activity is suppressed.
To begin with, when the research group compared a rat with neurological damage to a normal rat, they found that in the rat with neurological damage NPY nerve activity was weakened, and it experienced pain from tactile stimulus. In other words, allodynia symptoms were displayed as a result of the nerve damage. In light of that, when an optogenetic method was used to suppress only the NPY nerve activity in the spinal cord dorsal horn of the normal rat, allodynia symptoms appeared. Additionally, when the group increased NPY nerve activity in the rat with neurological damage, nerve activation in the region that senses pain from tactile stimulus was suppressed.
Essentially therefore, the group found that the decline in NPY nerve activity that follows nerve damage is the cause of neuropathic allodynia, and that conversely, the allodynia symptoms can be repressed by stimulating NPY nerve activity.
Tsuda says that "With this research we were able to newly discover a group of nerve cells that is important to the onset of neuropathic allodynia, for which even morphine is ineffective. Going forward we hope to reveal the mechanism behind this lowering of nerve cell activity. Furthermore, we hope to use rats to find a medicine that heightens NPY nerve activity from within the library of existing medicines, and move ahead with a physician-led clinical trial in order to come up with an effective medical remedy."
This article has been translated by JST with permission from The Science News Ltd.(https://sci-news.co.jp/). Unauthorized reproduction of the article and photographs is prohibited.




