A joint international research team consisting of Assistant Professor Phuoc Duy Tran and Professor Akio Kitao of the Department of Life Science and Technology, School of Life Science and Technology, Tokyo Institute of Technology, researcher Adnan Sljoka from the Center for Advanced Intelligence Project (AIP), RIKEN, Professor Scott Prosser of the University of Toronto, Professor Roger Sunahara of the University of California San Diego, and others, has succeeded in clarifying the mechanisms that are the key to activating the human adenosine A2A receptor (A2AR) through a combination of fluorine-19 nuclear magnetic resonance, molecular dynamics simulations, and mathematical rigidity theory.
(Credit: Tokyo Institute of Technology)
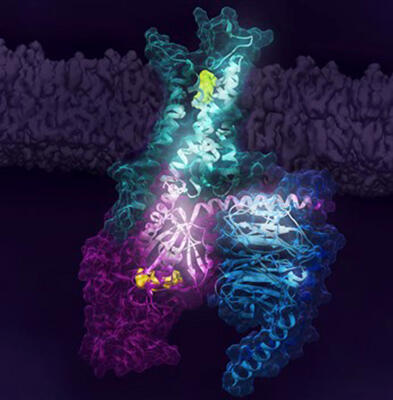
Human A2AR is a type of G protein coupled receptor (GPCR), which has the function of binding with G proteins and initiating cell signaling. It is a target molecule for a variety of drugs, including drugs for cancer and arteriosclerosis, but the activation process for A2AR, important for pharmacological phenomenon, was not well clarified.
Thus, the research team first investigated the NMR spectra changes caused by the G protein and ligand binding with A2AR using fluorine-19 nuclear magnetic resonance. As a result, they learned that at least two inactive states and three active states exist at the same time in A2AR, and their coexistence ratio changes depending on ligand binding and the activation state of G proteins.
Next, the team identified the long-distance signaling pathways to the G proteins from the A2AR-ligand binding sites by analyzing the complex spatial structures of A2AR and G proteins, obtained via molecular dynamics simulations, using mathematical rigidity theory. They were able to ascertain that the GβY subunits of a G protein trimer are particularly important to signaling and the complete activation of receptors.
Opportunities to develop new drugs
Kitao says that, "In the future, if we can clarify the differences of signaling pathways and intermediate states caused by dependence on acting molecules, we will be able to minutely control their functions by binding a variety of molecules to A2AR; we expect this to lead to opportunities to develop new drugs."
■Ligands: molecules that bind to specific biological molecules such as receptor proteins and have some sort of effect on them.
This article has been translated by JST with permission from The Science News Ltd.(https://sci-news.co.jp/). Unauthorized reproduction of the article and photographs is prohibited.




