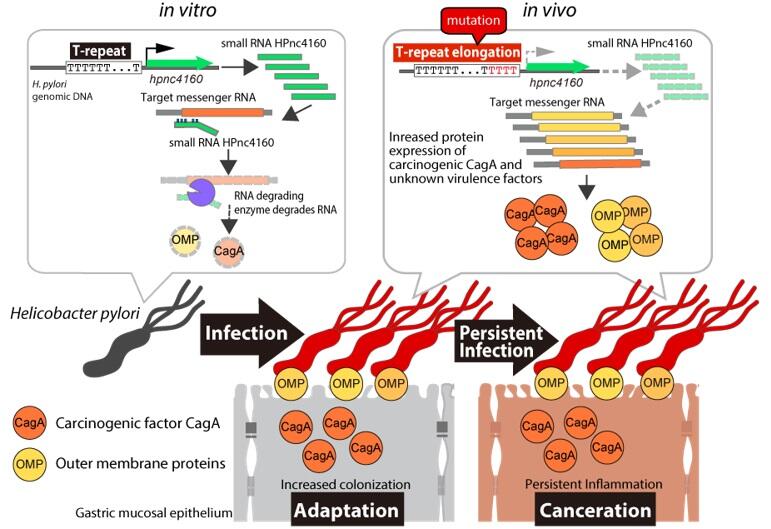
The research group led by Assistant Professor Ryo Kinoshita and Associate Professor Hitomi Mimuro at the Research Institute for Microbial Diseases at Osaka University discovered the mechanism by which H. pylori, often transmitted in early childhood, persistently infects the stomach for a long period of time and causes gastritis and gastric cancer decades later. They comprehensively analyzed the genomic DNA of H. pylori isolated from infected animals, including mice, and found that an sRNA, HPnc4160, which is important for the persistent infection, regulates the expression of pathogenic factors, such as CagA (a carcinogen associated with gastric cancer), leading to long-term infection. Their findings, published in the British scientific journal Nature Communications, are expected to lead to the diagnosis of H. pylori infection and the development of therapeutic methods.
Approximately half of the world's population and more than 60 million individuals in Japan are infected with H. pylori. In many cases, infection with H. pylori through beverages or foods in early childhood leads to a lifelong persistent infection of the gastric mucosa, causing atrophic gastritis, ulcers, and gastric cancer in the middle age, decades later. However, only a small proportion of individuals infected with H. pylori develop gastric cancer (approximately 0.4% of infected individuals per year), and most cases are of asymptomatic gastritis.
The genes of H. pylori are easily mutated, and each strain isolated from infected people has a unique genome sequence. Therefore, the genes of H. pylori are thought to acquire mutations in the stomach after infection, adapting to the changes in the stomach of the host, but the details remain unknown. The research group previously showed that CagA, a protein involved in the development of gastric cancer and the malignant transformation of gastritis, activates transcription factors of the host and suppresses apoptosis, allowing the bacteria to adhere to gastric cells for a long period of time.
In the present study, they comprehensively examined the mutations that occur in the genome of H. pylori after infection in vivo. Specifically, they inoculated H. pylori into the stomachs of mice (40 strains) and gerbils (eight strains) and performed whole-genome analysis on the bacteria isolated 8 weeks after the inoculation.
Credit: RIMD, Osaka University.
Results showed that mutations frequently occur in certain regions of its genomic DNA, and comprehensive analysis of the expression of RNAs encoded by the genes located around these regions revealed a particularly large variation in expression of the sRNA Hpnc4160. Moreover, successive thymidine (T) bases (T repeats) were found in the control region upstream of the gene encoding this sRNA. sRNAs are known to bind to messenger RNAs via complementary sequences and regulate the level of their translation into proteins.
Elongation of the T-repeat sequence was observed 8 weeks after infection, and the expression level of Hpnc4160 decreased with an increase in T repeat length. The research group then generated strains of H. pylori with different T repeat lengths, and their inoculation into mice resulted in different expression levels of Hpnc4160, depending on the T repeat length.
Next, they comprehensively analyzed the proteins whose expression is regulated by Hpnc4160 at the RNA and protein levels. It was found to target eight factors, including the genes encoding CagA, a carcinogenic factor in gastric cancer, and outer membrane proteins.
In addition, they examined the effect of Hpnc4160 on the pathogenicity by inoculating mice with the Hpnc4160-deficient mutant strain or the wild-type strain of H. pylori. Results showed that the number of viable bacteria was higher in the mice inoculated with the mutant strain 3 days after infection, when the T repeat elongation was not observed. In addition, the pathogenic factors, which are normally suppressed by Hpnc4160, increased the colonization of the bacteria in the mouse stomach. On the other hand, in the post-infection examination up to 4-56 days, the number of viable bacteria in mice inoculated with the Hpnc4160-deficient strain decreased 56 days after inoculation, when the T repeat elongation was observed. Moreover, a comparison of the viable bacteria with different T repeat lengths showed that the longer the T repeat, the more likely the strain persistently infected the stomach.
Additionally, the variation in T-repeat sequence increased over time after infection. This variation appears to be useful for adapting to the environment of the stomach, which changes depending on the infection status.
Furthermore, their examination of these characteristics in H. pylori isolated from patients with gastric cancer revealed long T-repeats and a tendency of increased expression of the Hpnc4160 target factors.
Therefore, the expression of Hpnc4160 in H. pylori suppresses pathogenic factors in vitro. However, the infection of the stomach decreases its expression due to elongation of the T repeat by gene mutation, and this increases the expression of pathogenic factor proteins, making it easier to persistently infect the gastric mucosa. If the diagnosis of highly pathogenic H. pylori infection that leads to gastric cancer is possible, excessive administration of antibiotics, which increases the occurrence of antibiotic-resistant bacteria, can be avoided.
Associate Professor Mimuro said, "Since we have now identified the pathogenic factors, we aim to investigate their functions in the future. I hope that understanding these will lead to the development of effective diagnostic and therapeutic agents."
This article has been translated by JST with permission from The Science News Ltd.(https://sci-news.co.jp/). Unauthorized reproduction of the article and photographs is prohibited.




