Like humans, even small insects have a brain and heart, but how are the cells that maintain such small organisms different from ours? The research group consisting of PhD student Akifumi Shiomi (currently RIKEN Research Fellow), Assistant Professor Kohjiro Nagao, Professor Emeritus Masato Umeda, and others at the Graduate School of Engineering at Kyoto University analyzed the physical properties of Drosophila cells, which are approximately 3 millimeters in length, and found that they were extremely flexible compared with mammalian cells and did not break even when a strong force was applied. They also found that the movement of lipids within the thin cell membrane with a thickness of only several nanometers regulated the hardness of cells, which is important for tissue formation and cellular function. These findings were published in Cell Reports.
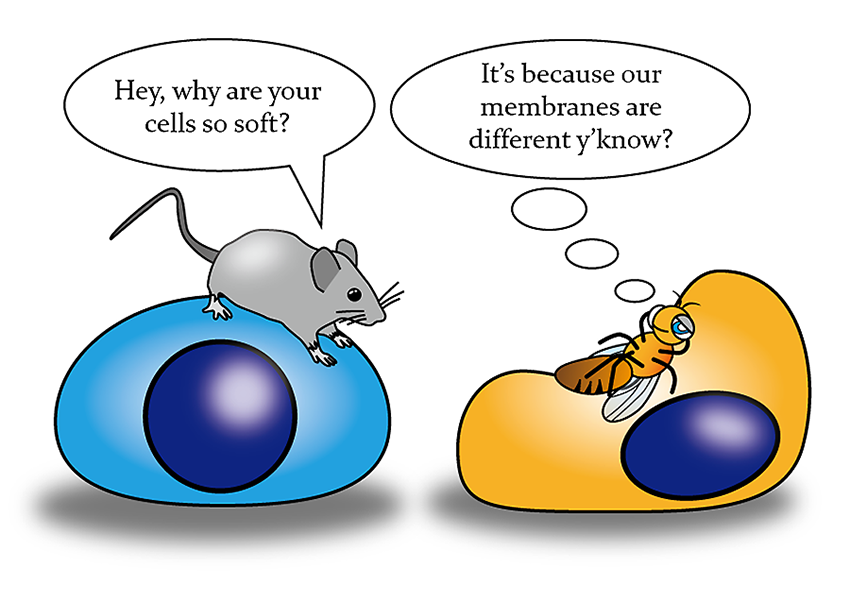
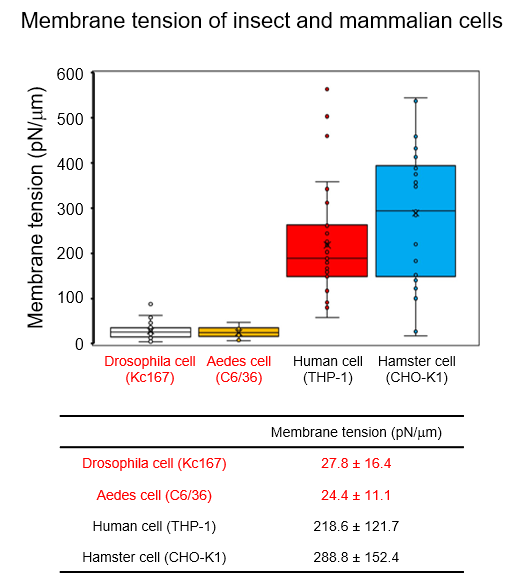
The research group evaluated the hardness of the cells of various species using the micropipette aspiration technique. They showed that Drosophila cells had significantly higher deformability and flexibility than mammalian cells.
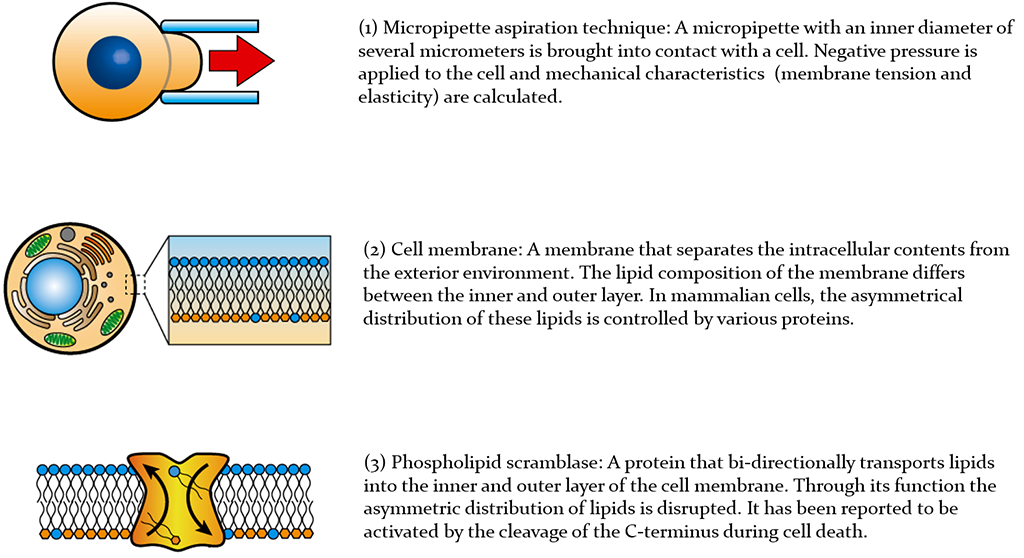
Next, they focused on the structure of the lipid bilayer that makes up the cell membrane surrounding the cell. In mammals, each layer of the lipid bilayer is composed of different types of lipids, and the asymmetric distribution of lipids is maintained by the action of various proteins. However, they found that the asymmetric distribution of lipids was disrupted in Drosophila cells and that the cause of this involved the phospholipid scramblase (XKR), which transports lipids bidirectionally between the two layers of the lipid bilayer.
The mammalian XKR protein functions only in special circumstances, such as during apoptosis. However, the research group found that Drosophila XKR also functions in living cells, always transporting lipids between the two layers of the lipid bilayer of the cell membrane. Additionally, the deformability of the cell membrane is mainly determined by the hardness of the lipid bilayer membrane and the cytoskeleton that supports it. Both these factors were affected by XKR, increasing the flexibility of Drosophila cells.
Furthermore, the suppression of XKR function in Drosophila hemocytes (blood cells) reduced the flexibility of the cells and their number circulating in the body. For a comparison, the research group allowed the insect XKR to function in mammalian cells, which resulted in the enhanced deformability of the cells. Thus, regulation of the hardness of cells by phospholipid scramblase may be a universal method for ensuring cell integrity and function.
Credit: Kyoto University
It has previously been reported that even in mammals, lipid scramblase may function in some cancer cells and blood cells. It is known that the hardness of cells in our body changes as a result of aging and diseases, such as cancer, and that the cell hardness influences various cell functions. The findings of this study are expected to contribute to our understanding of the pathophysiology of various diseases as well as their treatment.
Unlike the Drosophila and mosquito cells examined in this study, the hardness of Spodoptera frugiperda cells, which are relatively longer, was comparable to that of mammalian cells. In the future, by analyzing the deformability of cells and the function of XKR in various insects of different sizes, the research group aims to determine whether increasing the deformability of the cells contributes to the miniaturization of insects.
As explained by Assistant Professor Nagao, "Our discovery that cell deformability is enhanced by the movement of lipids within the thin cell membrane with a thickness of several nanometers was unexpected. To demonstrate our discovery, we needed to evaluate the movement of lipids and the hardness of cells using various analytical methods, and we were able to achieve this research goal with the support of many collaborators. Although working on this research project involved a series of trials and errors, we have gained valuable experience as researchers by learning various experimental methods and concepts, which will contribute to our future research work."
This article has been translated by JST with permission from The Science News Ltd.(https://sci-news.co.jp/). Unauthorized reproduction of the article and photographs is prohibited.




