A research group including Graduate Student Kazuki Murakami, Associate Professor Shinji Kajimoto, Professor Takakazu Nakabayashi, and others from the Graduate School of Pharmaceutical Sciences at Tohoku University has successfully developed a technique for evaluating the liquid-liquid phase separation of proteins believed to be associated with the onset of neurodegenerative diseases.
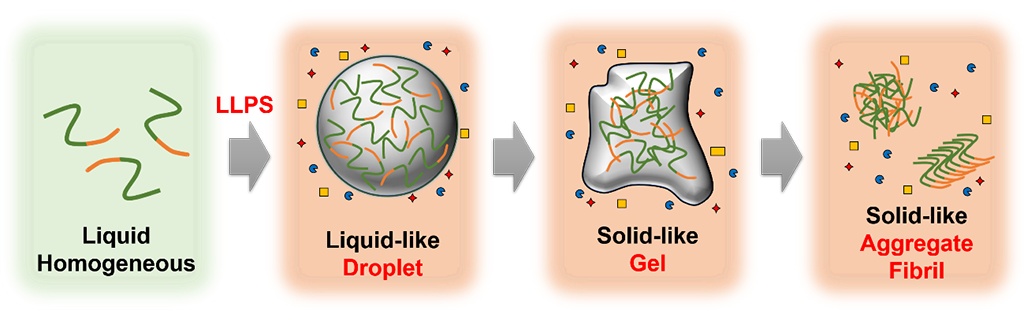
Even though no radical treatment is available yet, a model of neurodegenerative diseases suggests that highly concentrated phases of proteins (droplets) generated by liquid-liquid phase separation (LLPS) produce protein fibrils that induce disease onset. However, the method for investigating these droplets while maintaining their original phases, remains unavailable, and therefore, the details of these droplets are poorly understood.
The research group investigated the structure and concentration of proteins in droplets in their original phases using Raman microscopy, which measures light scattering to evaluate molecular structure and concentration. Professor Nakabayashi said, "Accurate measurement of Raman scattering intensity is necessary to determine protein concentration in droplets, and it took some ingenuity to quantitatively measure the light scattering intensity under a microscope."
This study revealed, for the first time, that ataxin-3, which causes the neurodegenerative disorder Machado-Joseph Disease, induces LLPS. Furthermore, the concentration and structure of ataxin-3 in its original phase were evaluated in droplets. It was vital to focus on water, which was measured simultaneously with the proteins. Since the number of water molecules is extremely large, the concentration of water can be regarded constant. Therefore, using the Raman scattering intensity of water as an intensity standard, the Raman scattering intensity of proteins in droplets could be quantified accurately.
Credit: K. Murakami et al. Chem. Sci. 12, 7411-7418 (2021) licensed under CC by-NC 3.0
This demonstrates that the protein concentration in LLPS-generated droplets can be measured accurately and that the protein concentration in these droplets is affected by the properties of the surrounding solution. This finding indicates that concentrations of proteins in intracellular droplets are affected by the surrounding intracellular environment. The protein droplets produced inside cells return to the uniformly dispersed original phases, but some droplets are believed to transform into protein aggregates that induce neurodegenerative diseases. In this study, the research group demonstrated that highly concentrated droplets can form protein aggregates and proposed a model that explains how abnormal intracellular conditions contribute to the formation of aggregates.
Professor Nakabayashi says, "We plan to use this technique to measure LLPS associated with various neurodegenerative diseases and determine the concentrations, structures, and interactions of proteins present within the droplets, which will help clarify the pathogenesis of neurodegenerative diseases."
This article has been translated by JST with permission from The Science News Ltd.(https://sci-news.co.jp/). Unauthorized reproduction of the article and photographs is prohibited.




