The kidney has multiple nephrons arranged at the tips of multi-branched collecting ducts, which is very similar to the arrangement of a broccoli sprig. To date, construction of this higher-order structure solely from embryonic stem (ES) cells and induced pluripotent stem (iPS) cells has been challenging. A research group led by Professor Ryuichi Nishinakamura, lecturer Shunsuke Tanigawa, and graduate student Etsuko Tanaka of the Institute of Molecular Embryology and Genetics at Kumamoto University has succeeded in creating a higher-order kidney structure generated from ES cells alone. Furthermore, after transplanting the prepared kidney tissue into mice models, it was confirmed that the blood vessels therein penetrated tissue and matured properly. This is a major step toward development of next-generation regenerative medicine, wherein kidney structures can be artificially produced outside the body for transplantation. These findings were published in the online version of Nature Communications.
The kidneys play an important role in life support by producing urine; however, they do not regenerate if damaged. There are more than 13 million patients with chronic renal failure in Japan, of whom 340,000 are on dialysis; however, kidney transplantation is extremely limited due to shortage of kidney donors. The kidney contains approximately one million nephrons in adults and 10,000 nephrons (glomeruli and tubules) in mice, which are connected by the tips of collecting ducts. The interstitium fills the space between these nephrons and collecting ducts and consists of stromal progenitors. Interstitial progenitor cells play a role in controlling nephron differentiation and ureteric bud branching. Without this function, higher-order structures in the kidney cannot be formed.
In 2014, the research group established a method for inducing nephron progenitor cells from mouse ES cells and human iPS cells, and in 2017, they proposed a method for inducing ureteric buds. In this study, they developed a method for inducing residual stromal progenitor cells. Initially, the gene cluster characteristic for interstitial progenitor cells and the developmental mechanism were identified using a fetal mouse kidney at 11.5 days of gestation. Next, using antibodies and a culture condition, they established a method to isolate the posterior intermediate mesoderm to induce stromal progenitors. These steps were instrumental in developing a method to derive stromal progenitor cells from mouse ES cells via the posterior intermediate mesoderm. The induced interstitial progenitor cells were cultured in vitro in combination with nephron progenitor cells and ureteric buds, which were also derived from ES cells. In doing so, they succeeded in creating the original higher-order structure of the kidney, wherein nephrons were placed around a multi-branched collecting duct.
When this kidney tissue was transplanted into immunocompromised mice, blood vessels penetrated and matured further. Specialized stromal cells, such as mesangial cells, which are important for filtration, and renin-producing cells, which are responsible for blood pressure regulation, also differentiated from ES cells. Thus, they successfully constructed a fully pluripotent stem cell-derived kidney with a higher-order structure by establishing a third (stromal progenitor) induction method that was essential for nephrogenesis. Lecturer Tanikawa said, "The number of transplanted organoid nephrons was about 50 to 100, and we suspect that another factor is needed to be considered to increase and mature this number. In addition, since the ureter that connects to the urethra is not formed, urine-like substances are accumulated in the collecting ducts. In the future, we would like to identify factors for further maturation while forming a ureter that extends outside the kidney." Professor Nishinakamura said, "Fetal kidneys cannot be used for research the same way as mice ones, so, we will use information about various gene expression published in databases. By doing so, I believe that we will be able to create higher-order structures in human iPS cell-derived kidneys by the next year."
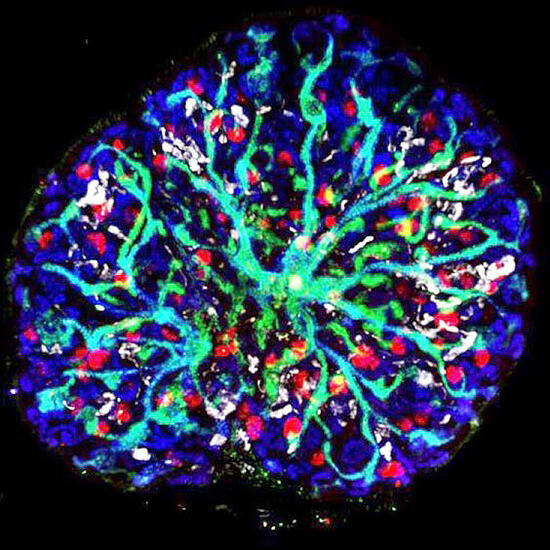
(Provided by Professor Ryuichi Nishinakamura, Kumamoto University) All rights reserved
This article has been translated by JST with permission from The Science News Ltd.(https://sci-news.co.jp/). Unauthorized reproduction of the article and photographs is prohibited.




