An Ehime University Graduate School of Science and Engineering research group consisting of Doctoral researcher Fan Wu (currently a researcher at Nanjing University), Associate Professor Masayoshi Takase, and Professor Hidemitsu Uno, in collaboration with Professor Zhen Shen of the School of Chemistry and Chemical Engineering, Nanjing University, succeeded in synthesizing wing-shaped nitrogen-doped nanographene.
DOI: 10.1021/acs.orglett.1c03669
Provided by Ehime University
In recent years numerous synthetic chemical approaches have been investigated using polycyclic aromatic compounds (PAH) which are regarded as highly pure nanographenes that have a clear molecular structure. If it becomes possible to construct structurally controlled three-dimensional aggregates and spaces from these two-dimensional nano-sized graphenes, then it is expected that they will create new materials with properties that are based on these unique structures. The research group has been conducting studies on using pyrrole (a type of heterocyclic aromatic compound amine) to synthesize hexapyrrolohexaazacoronenes (HPHACs), which are nitrogen-doped PAHs, and on clarifying their physical properties.
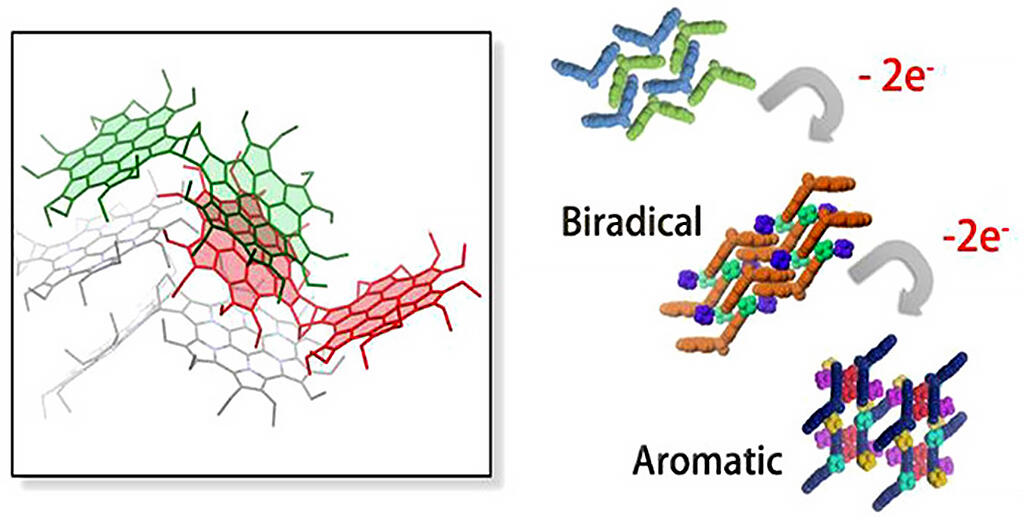
HPHACs, which are composed of electron-rich pyrroles, are easily oxidized, and their two-electron oxidants have unique characteristics, such as global aromaticity. Previous research has only focused on the synthesis of analog-containing monomers and on clarifying their property correlations, and no studies have been conducted on dimer synthesis aimed at constructing structurally controlled three-dimensional structures.
In this research, a bicyclooctadiene skeleton frame was adopted as a structurally rigid cross-linking site, and wing-shaped, bent HPHAC dimers were synthesized. Similarly to known HPHAC monomers, stable redox properties were observed in the newly synthesized dimers. Additionally, the integration modes according to oxidation state were clarified via single crystal structure analysis and a variety of spectroscopic measurements.
Furthermore, it revealed that, in tetracation state HPHAC dimers, the aromaticity of the three-dimensional space surrounded by two HPHACs is increased. This indicates that the magnetic and electronic properties of the HPHAC are the result of them influencing each other.
According to Associate Professor Takase, "By using redox-active nanographene for wings, it has been possible to construct a variety of aggregates according to the oxidation states, and in recent years the synthesis of curved π-electron compounds, etc., has been achieved. On the other hand, there are few examples of constructing structurally controlled three-dimensional aggregates and spaces that use them as constituent units. If high mechanical robustness and large surface area can be realized, then we expect that they can be applied to sensors, imaging, and energy transformation materials, and so on."
This article has been translated by JST with permission from The Science News Ltd.(https://sci-news.co.jp/). Unauthorized reproduction of the article and photographs is prohibited.




