The so-called "Moderna arm" is a delayed, large-scale, and localized reaction that causes redness, swelling, pain, and itching around the vaccination site approximately seven days after being vaccinated with Moderna's mRNA-1273 COVID-19 vaccine. This reaction is commonly called "COVID arm" in the United States and "Moderna arm" in Japan. This adverse reaction in the skin has very different clinical characteristics from allergic reactions to conventional drugs and vaccines, and little is known about how it occurs.
A research group led by Dr. Toshihide Higashino, Director of the Department of Dermatology at the Self-Defense Force Central Hospital, conducted a research study on the incidence rate of Moderna arm, interviewing people who received the mRNA-1273 vaccine for their first vaccination and then inferring whether or not they had delayed, large-scale, and localized reactions after vaccination. Through the results, it was found that this adverse reaction occurred more frequently than was previously thought. It was also found that it is more likely to occur in women, and in the 30s to 60s age group.
The research group conducted a cross-sectional study on the incidence rate of delayed, large-scale, and localized reactions by inferring whether an individual had developed a delayed, large-scale, and localized reaction after vaccination based on the content of second vaccination pre-vaccination interviews for individuals who received the mRNA-1273 vaccine for their first vaccination shot. In the epidemiological study through these interviews and with the approval of the Ethics Committee of the Self-Defense Forces Central Hospital, five dermatologists interviewed a total of 5,893 vaccinated individuals (3,318 men and 2,575 women) from May 24, 2021, to November 30, 2021, at the Self-Defense Forces large-scale vaccination center in Otemachi, Tokyo.
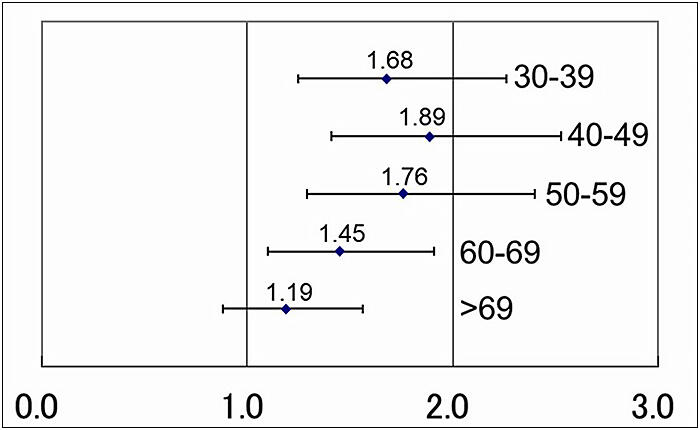
The study referred to previous reports on delayed, large-scale, and localized reactions and assumed that a delayed, large-scale, and localized reaction had occurred if the individual was aware of symptoms such as redness, swelling, hardened skin, itching, or burning around the vaccination site from six days after vaccination. The results showed onset in 747 individuals (12.7%). The incidence rate in the study was significantly higher than in similar studies that had been previously conducted, and it was found that this adverse reaction frequently occurs after vaccination, including for individuals without strong subjective symptoms. On the other hand, none of the subjective symptoms considered to be delayed, large-scale, and localized reactions were severe enough to be considered contraindications to vaccination. The incidence rate in women was 22.4% (577 out of 2,575 individuals) and in men was 5.1% (170 out of 3,318 individuals), which is a significant difference, with women approximately 5.3 times more likely to develop it than men. The incidence rate for individuals under the age of 30 was 9.0%, which tended to be lower than for individuals in their 30s (14.3%), 40s (15.8%), 50s (14.9%), and 60s (12.6%).
Assessment of Delayed Large Local Reactions After the First Dose of the SARS-CoV-2 mRNA-1273 Vaccine in Japan Toshihide Higashino, Yuki Yamazaki, Satoko Senda et al. JAMA Dermatol. Published online June 1, 2022.
doi:10.1001/jamadermatol.2022.2088
Dr. Higashino said that "Except for cases in which symptoms had resolved by the third day after vaccination, we carefully interviewed the patients at the vaccination site about adverse skin reactions, and even identified cases that were not recognized by the patients themselves as adverse reactions. In the future, we plan to continue with similar cross-sectional studies on adverse reactions after the second and subsequent COVID-19 vaccinations in order to understand adverse reactions that occur less frequently."
This article has been translated by JST with permission from The Science News Ltd.(https://sci-news.co.jp/). Unauthorized reproduction of the article and photographs is prohibited.




