A joint research group consisting of members of Tokyo University of Pharmacy and Life Sciences, Chiba University, Kumamoto University, Ono Pharmaceutical and Eisai have developed a human multicellular spheroidal blood-brain barrier model with a structure that imitates the in vivo blood-brain barrier by combining three types of independently created reversible immortalized cells in a hierarchical manner. It is hoped that this will contribute to the creation of new drugs for central nervous system diseases, as it makes it possible to assess whether antibody drugs and peptide drugs reach the human brain. In addition to this being published in Molecular Pharmaceutics, the details of the associated protocols were also published in Bio-protocol.
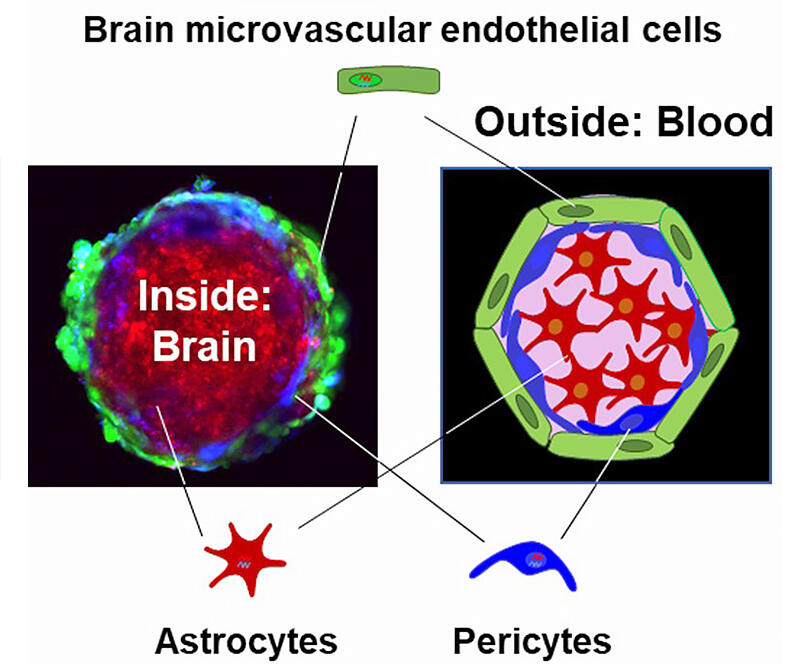
Left, a picture showing the hiMCS-BBB model structure; right, an illustration of the models. Astrocytes form a spheroid core, over which pericytes accumulate. Brain microvascular endothelial cells cover the astrocyte-pericyte spheroid to form a layer serving the BBB. Thus, while in an inverted manner (the inside is designated as "brain" whereas the outside is designated as "blood"), the hiMCS-BBB models structurally mimic an in vivo BBB. The picture was taken by a confocal microscope after fluorescent dye labelling (green, brain microvascular endothelial cells; blue, pericytes; red, astrocytes). The picture and illustration are cited from Kitamura et al., Mol Pharmaceut 2022
doi: 10.1021/acs.molpharmaceut.2c00120 with slight modification.
The research group created a 3D human blood-brain barrier model that mimics the in vivo blood-brain barrier by positioning three types of human reversible immortalized blood-brain barrier cells (HBMEC/ci18, HASTR/ci35, HBPC/ci37) in a layered and hierarchical manner. The model is an inverted (inside-out) version of the in vivo blood-brain barrier; it has been named the human immortalized cell-based multicellular spheroidal blood-brain barrier (hiMCS-BBB) model. The group confirmed that this model has the essential functions of the blood-brain barrier, including a barrier created by intercellular binding and excretion transporter functions, as well as transferrin and insulin transcytosis (receptor-mediated transcytosis).
The group carried out permeability experiments using antibodies and peptides to evaluate the performance of the hiMCS-BBB model as a way of evaluating the ability of antibodies and peptides to move in the human brain. When assessing antibodies, the group used the anti-transferrin receptor antibody MEM189, which is thought to penetrate the blood-brain barrier, and the anti-transferrin receptor antibody 13E4, which is believed not to penetrate the blood-brain barrier. When evaluating peptides, they used SLS peptide, which does penetrate the blood-brain barrier, and DNP peptide, which does not. The results indicated that MEM189's blood-brain barrier permeability is higher than that of 13E4, and, similarly, SLS peptide's blood-brain barrier permeability is higher than that of DNP peptide.
The hiMCS-BBB model developed by the group can be created ad infinitum. This study was also able to verify its characteristics and usefulness by repeatedly creating the model to carry out a variety of experiments. Thus, this model is not only able to screen antibodies and peptides; it also enables experiments to be carried out in stable conditions that can be repeated in a short space of time. It is hoped that this will become a new basic technique for drug discovery that will accelerate the development of antibody drugs and peptide drugs for central nervous system diseases.
This article has been translated by JST with permission from The Science News Ltd.(https://sci-news.co.jp/). Unauthorized reproduction of the article and photographs is prohibited.




