First-year doctoral student Kosuke Ono and Professor Masaru Shimomura from the Graduate School of Science and Technology, Shizuoka University , in collaboration with the Nagoya Institute of Technology and the University of Peradeniya in Sri Lanka, have successfully grown anatase-type titanium oxide epitaxial thin films by the solvothermal method. Fabrication of the thin film is less than one-tenth the cost of the vapor phase method. This is expected to lead to research on the interrelationship between the physical properties of photocatalysis and doping, as well as the development of devices and solar cells that make use of photocatalysis. The group's research was published in Chemical Engineering Journal.
Provided by Shizuoka University
Titanium dioxide has multiple crystal structures, including rutile, anatase, and brookite types. Among these, single crystals and rutile type epitaxial thin films are easy to produce as there are established methods. However, single crystals for anatase and brookite types are limited to natural materials, and while methods for producing epitaxial thin films exist, they are expensive. Epitaxial thin films have many electrical and optical advantages over polycrystalline thin films and will pave the way for new applications to be created and also help improve current ones if they can be easily fabricated.
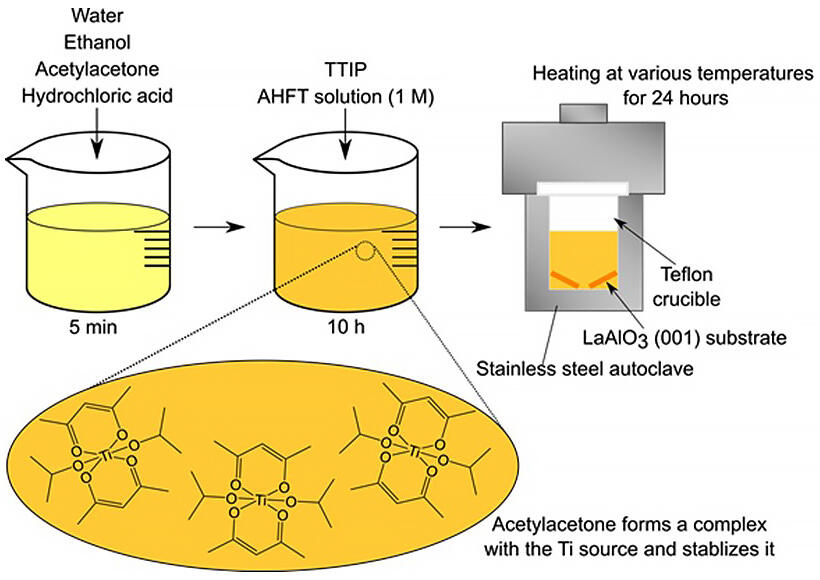
In conventional titanium dioxide preparation methods using the solvothermal method (a method to synthesize substances under high temperature and high pressure by sealing solutions containing raw materials or solid raw materials and solvents in a metal pressure-vessel and heating them), synthesis has been performed with solutions in which hydrochloric acid makes up half of the total solution volume. However, lanthanum aluminate single-crystal substrates used for the preparation of anatase-type titanium dioxide epitaxial thin films are corroded by acid under high temperature and high pressure in the solvothermal method. At the same time, hydrochloric acid functions as a stabilizer for the titanium source reagent, and if the amount used is reduced, the titanium source reagent, which is highly reactive with water, will decompose before the thin film is formed.
The research group used acetylacetone as a stabilizing agent instead of hydrochloric acid to form an organometallic complex based on titanium, stabilized the titanium ions in the solution, suppressed the corrosion of the substrate, and improved the solvothermal method to successfully fabricate epitaxial thin films of anatase-type titanium oxide by the solvothermal method.
Specifically, they prepared a solution by adding a titanium source reagent and a fluorine compound for growth control to a solvent based on water, ethanol and acetylacetone, while reducing the amount of acid used. Using this solution, the research group established a new and simple method to prepare anatase-type titanium dioxide epitaxial thin films by the solvothermal method. They also succeeded in achieving both thin film growth and suppression of the corrosion of the lanthanum aluminate single-crystal substrate, which had been a problem.
Furthermore, they prepared thin films doped with the rare earth element erbium and conducted X-ray fluorescence holography (XFH). XFH revealed that the erbium atoms introduced into the anatase-type titanium oxide substituted the titanium atoms. It has been reported in a number of cases that the addition of rare earth elements and transition metal elements to anatase-type titanium dioxide changes its physical properties, including photocatalysis. This result is a pioneering achievement for future experimental studies of the relationship between the change in its physical properties and dopants from the viewpoint of structural change.
"It was a struggle to achieve both the suppression of substrate corrosion and the growth of the target thin film," says Kosuke Ono. "Moving forward, we intend to further analyze structural analysis data to have a better understanding of the relationship between photocatalysis and dopants. We will also study new applications."
This article has been translated by JST with permission from The Science News Ltd.(https://sci-news.co.jp/). Unauthorized reproduction of the article and photographs is prohibited.




