A research group led by Associate Professor Hirokazu Kobayashi of the Komaba Institute for Science, Graduate School of Arts and Sciences at The University of Tokyo, Professor Atsushi Fukuoka and graduate student (at the time of research) Yusuke Suzuki of the Institute for Catalysis at Hokkaido University, Assistant Professor Takuya Sagawa of the Faculty of Engineering at Tokyo University of Science, and project leader Makoto Saito of the Basic Chemicals Division at Showa Denko have succeeded in efficiently synthesizing chitin-oligosaccharides by hydrolysing chitin obtained in large quantities from crab shells.
Provided by the University of Tokyo
Chitin-oligosaccharides, which are obtained by hydrolyzing the abundant chitin contained in crab shells, have been found to bring out the immunity of plants. If these compounds can be used in agriculture, it could reduce the risk of crops becoming infected with fungi and other organisms, even with low amounts of pesticides used, and could lead to higher yields. However, chitin is difficult to break down and it has been a challenge to selectively synthesize chitin-oligosaccharides.
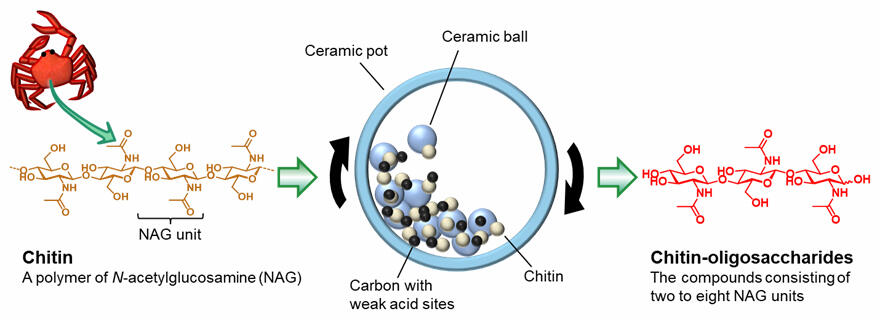
Chitin is a macromolecule consisting of numerous N-acetylglucosamine (NAG) sugars, which can be gradually reduced to smaller molecules by cleaving the bonds between the sugar units, which allows chitin-oligosaccharides with two to eight NAGs to be obtained. The research group came up with the idea that solid chitin could be hydrolyzed by activating the chitin molecules with mechanical force and then using a catalyst to attack the activated chitin molecules.
Based on this idea, the researchers used a grinding machine called a ball-mill to apply mechanical force in the hydrolysis reaction of chitin. They conducted the reaction by placing a solid catalyst, chitin and ceramic balls in a ceramic pot and setting the pot in the grinding machine and operating it at a speed of 500 RPM. After testing various catalysts, they found that a carbon material exhibiting weak acidity acted as a catalyst to efficiently hydrolyze chitin and produce chitin-oligosaccharides. Normally, minerals that exhibit strong acidity are used as catalysts for reactions that break bonds between sugars, but the fact that the weak acidity of the carbon material showed higher activity and selectivity was an important discovery.
Analysis of the reaction showed that the larger chitin molecules were hydrolyzed faster to produce the desired smaller oligosaccharides as products, whereas the reaction that further degraded the smaller oligosaccharides was suppressed. This results in chitin-oligosaccharides with high selectivity. Carbon powerfully adsorbs large chitin molecules while only weakly adsorbs small oligosaccharides, which the researchers believe may have also contributed to the improved selectivity.
"In our research, we added activated carbon, which is weakly acidic to chitin, as a catalyst and applied the mechanical force of ball milling to hydrolyze chitin, and it became clear that this produced chitin-oligosaccharides with high selectivity," explains Kobayashi. "We hope to put this method to practical use to contribute to pesticide-free agriculture."
Journal Information
Publication: Angewandte Chemie International Edition
Title: Selective Synthesis of Oligosaccharides by Mechanochemical Hydrolysis of Chitin over a Carbon-Based Catalyst
DOI: 10.1002/anie.202214229
This article has been translated by JST with permission from The Science News Ltd.(https://sci-news.co.jp/). Unauthorized reproduction of the article and photographs is prohibited.




