Brain diseases such as Alzheimer's and Parkinson's are thought to be caused by the deposition of abnormal proteins in the brain, which lead to motor impairment and cognitive decline. Imaging technologies have been established for tau protein lesions, a core pathology in Alzheimer's disease. However, abnormal aggregates of α-synuclein that cause Parkinson's disease are deposited less than tau lesions, so a technology for imaging them in the living brain has not yet been created.
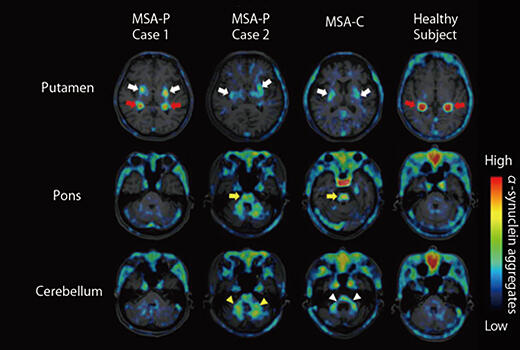
A research group led by Makoto Higuchi of the Quantum Life and Medical Science Directorate of the National Institutes for Quantum Science and Technology, in collaboration with three pharmaceutical companies (Eisai Co., Ltd., Ono Pharmaceutical Co., Ltd. and Takeda Pharmaceutical Company Co., Ltd.), has developed 18F-SPAL-T-06, a radioligand that visualizes the deposition and distribution of α-synuclein fibrils in the brain with high contrast. In this study, three patients with multiple system atrophy, who had Parkinsonian symptoms, and one healthy elderly person underwent brain imaging examinations using the group's newly developed agent. The results showed a high accumulation of 18F-SPAL-T-06 in the patients' deep brain regions. The location and amount of the radioligand accumulation were also consistent with the distribution of α-synuclein aggregates reported in previous studies.
Furthermore, a comparison of brain samples from a patient with multiple system atrophy and a healthy elderly subject confirmed that 18F-SPAL-T-06 bound strongly to α-synuclein aggregates, demonstrating its potential utility as a diagnostic tool. Imaging the deposition of these abnormal proteins in the brain, as achieved through this research, is expected to help elucidate the pathology of Parkinson's disease and related dementias, develop therapeutic drugs, and predict and prevent the onset of dementia.
Comparison of α-synuclein deposition images in patients with multiple system atrophy and a healthy elderly subject