Researcher Yuta Shudo, Chief Researcher Tohru Kawamoto and Senior Researcher Akira Takahashi of the Nanoparticle Functional Design Group, Nanomaterials Research Institute at the National Institute of Advanced Industrial Science and Technology (AIST), have developed a new adsorbent that can recover and concentrate methanol by improving the pigment Prussian blue.
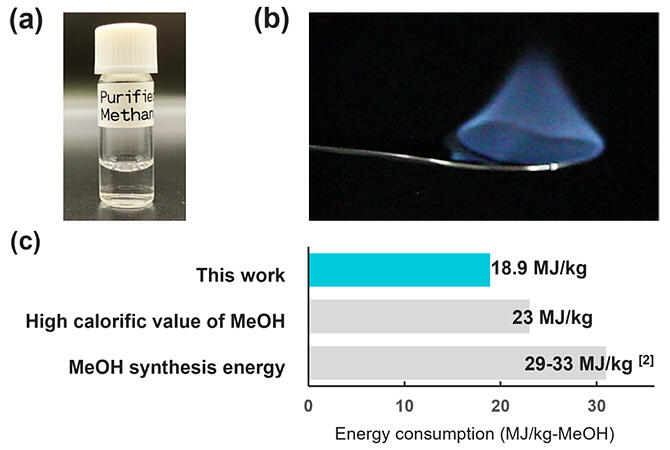
(b) photographs of actual combustion
(c) comparison of energy consumption attainted in this research
Provided by AIST
Methanol is widely used in chemical plants as a raw material for plastics and as a solvent. However, it causes air pollution such as fine particulate matter (PM2.5) when released into the atmosphere, so it is detoxified by combustion or other means. The main reason why methanol is not recovered as a resource is that when methanol is adsorbed, water vapor is also adsorbed at the same time during the recovery process as it has a high affinity for methanol. Due to the similar chemical properties of water and methanol, conventional adsorption and concentration techniques cannot selectively extract methanol.
This has led to adsorbent materials that can adsorb methanol more strongly than water to be developed. The researchers optimally designed the type and composition of the metal ions comprising the adsorbent Prussian blue to match the molecular structure of methanol, resulting in a methanol adsorption capacity five times higher than that of activated carbon. Water vapor, which is simultaneously adsorbed during methanol recovery, can be removed by heat treatment at 70 ℃ and separated from the more strongly adsorbed methanol. After the water vapor is removed, methanol with a high concentration of 95 wt% was able to be recovered as a liquid by heating it to 150 ℃.
The researchers hope that the new adsorbent will lead to the realization of a society that recycles resources, where organic solvents such as methanol, which has to be detoxified, can be reused as a resource.
"The challenge with this adsorbent is that it also adsorbs some water vapor, but we were able to recover it as a 95 wt% methanol liquid by making use of the different desorption temperatures for water and methanol," said Takahashi. "In the future, we would like to recruit partners to do joint research in methanol adsorption and concentration, as well as apply this to other alcohol molecules as a basic technology."
This article has been translated by JST with permission from The Science News Ltd. (https://sci-news.co.jp/). Unauthorized reproduction of the article and photographs is prohibited.




