Microbubble solutions, in which many 'microbubbles' ‐ air bubbles the size of a few tens of micrometers (one millionth of a meter) ‐ are dispersed in water, is used in familiar applications, such as carbonated water and Jacuzzi baths, in industry for fish and seafood aquaculturing and for cleaning semiconductor equipment. Bubbles in water normally rise and disappear at the surface, but nanobubbles, which are even smaller than microbubbles, have been reported to remain stable in water for weeks to months. However, there has been no theory to explain this phenomenon.
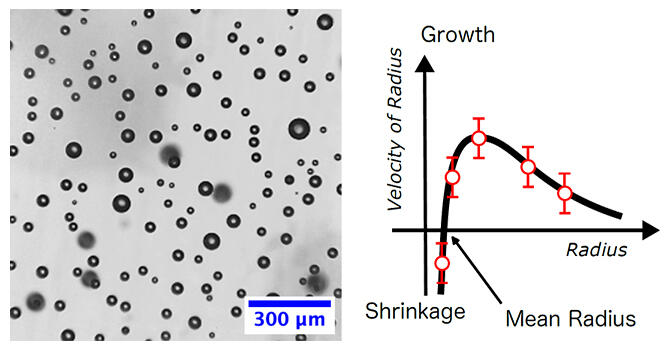
Using microscopically observable microbubbles, Associate Professor Yuki Uematsu at the Graduate School of Computer Science and Systems Engineering at the Kyushu Institute of Technology and his colleagues have identified the laws that predict the growth and shrinkage of individual bubbles. Specifically, they observed about 1,000 microbubbles under a microscope and took images every minute. They then analyzed the images to obtain the time variation of the radius of each bubble over a 90‐minute period. Their analysis revealed that at a certain point in time, bubbles larger than the mean radius expanded and bubbles smaller than the mean radius contracted. This is similar to 'Ostwald ripening,' where there are a large number of crystalline particles in a saturated crystalline solution, smaller particles become smaller and larger particles become larger. It is a phenomenon that can quantitatively predict and explain the behavior of microbubbles in water.
The research team is continuing their work, as their findings are key to improving methods for controlling the size of bubbles in microbubble water and to understanding the stability of nanobubbles. In the future, this could lead to contributions to multiple fields, such as the development of water for cleaning that removes dirt without detergents, or water for agriculture and fisheries that increases vegetable yields and higher survival rates in fish and shellfish aquaculture.