Among the various polymer materials used in daily life, only plastic bottles have achieved closed‐loop recycling and are reused without degradation. The production of polymers continues to increase, and predictions indicate that by 2050, the amount of waste derived from polymers will increase by over threefold compared with the current levels. A research group led by Associate Professor Daisuke Suzuki of the School of Science and Technology, Faculty of Textile Science and Technology at Shinshu University, has developed a material recycling method for polymer particles. By simply soaking the used polymer materials in a solvent, they can be broken down into their original fine particles and reused, resulting in low energy consumption and no degradation.
Suzuki said, "We have demonstrated the concept with polymethyl methacrylate, but similar recycling is possible with any polymer composed of fine particles. Statistics show that one‐fifth of the polymers produced in Japan are composed of microparticles. We are currently conducting joint research with the paint and tire industries, and this research was conducted in cooperation with SEKISUI CHEMICAL CO., LTD. We hope to use this method as a design guideline when creating new materials for the next generation." The result has been published in the journal Green Chemistry.
Provided by Shinshu University
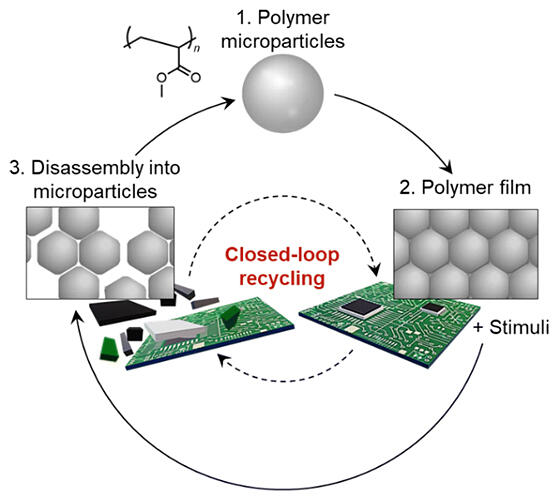
Polymer microparticles ranging in size from tens to hundreds of nanometers are used in cosmetics, paints, paper processing and other applications. By simply drying a dispersion containing these tiny microparticles, the microparticles can fuse together to form a particulate film. For this reason, they are used in adhesives and paints; however, this has poor strength.
Therefore, various chemical reactions can be induced at the fine particle interface using plasticizers, etc., to firmly bind the microparticles and strengthen them. However, once they are formed, the chemical bonds are difficult to break once again, often requiring special chemical reagents and harsh conditions such as high pressure and temperature. In addition, once the interfaces are forcibly broken in this way, recovering the material in a reusable state is challenging. Even if the material could be recovered, its quality would be degraded; therefore, the reuse of fine‐particle films while maintaining their high quality is considered difficult.
The newly developed method enables recycling by maintaining decomposition at the particulate level instead of the monomer level, as in the conventional method. Specifically, by simply placing the polymer film, or other material to be recycled, in a solvent containing a mixture of ethanol and water in a certain ratio, it can be degraded to the particulate level within 24 hours. The fine particles can be recycled by removing them from the mixture. The yield exceeds 99%, and the film produced after recycling performs at par with that before recycling.
What is particularly remarkable about this closed‐loop method is that films and adhesives bonded to various inorganic materials can be decomposed and easily separated from additives after the inorganic materials are removed. In these experiments, several particles measuring 57, 477, and 829 nanometers were recovered. Adhesives and paints used for semiconductor substrates can be recycled. Although such polymers are also used in the smooth parts of wrapping paper, they are difficult to separate and recover, even in Europe where recycling technologies are more advanced.
According to the university, the new method can separate the fine particles from paper, rendering their recycling possible. This approach, which can potentially solve the global polymer recycling problems, is a new low‐energy, high‐quality, and high‐efficiency recycling method that will change the prevailing concept of polymer recycling.
This article has been translated by JST with permission from The Science News Ltd. (https://sci-news.co.jp/). Unauthorized reproduction of the article and photographs is prohibited.




