A research group led by Assistant Professor Ichiro Yamauchi of the Graduate School of Medicine and Associate Professor Takuya Kubo of the Graduate School of Engineering at Kyoto University, in collaboration with Head Daisuke Nakajima of the Health and Environmental Risk Division and Deputy General Manager Shoji Nakayama of the Japan Environment and Children's Study (JECS) Programme Office at the National Institute for Environmental Studies (NIES), has announced that 3,3',5-triiodothyroacetic acid (TRIAC), a thyroid hormone analog found in effluents from sewage treatment plants, has endocrine-disrupting effects. The research group evaluated the effects of TRIAC by developing a mouse model in which the effects of thyroid hormones could be assessed. In vitro, TRIAC, which is equivalent to thyroid hormone triiodothyronine (T3) and thus has thyroid hormone activity, has been shown to reduce hormone action in the brain by suppressing hormone secretion. The findings are expected to lead to environmental research and health impact assessments. The results have been published in the June 21, 2023 issue of the international journal iScience.
Provided by KyotoU/Ichiro Yamauchi
The research group had previously found TRIAC, which is thyroid hormone analogue, in effluents from several sewage treatment plants. TRIAC is used as a therapeutic agent overseas but not in Japan. Overseas studies have reported the presence of substances that are thyroid hormone analogues in effluents from several sewage treatment plants, suggesting that TRIAC is produced via microbial biosynthesis.
The TRIAC compound has a different side chain to that of the endogenous thyroid hormone T3. Both have similar effects in vitro but reportedly behave differently in vivo, including the way they bind to their receptor. Because thyroid hormone is known to be essential for the metamorphosis of tadpoles into frogs, its effects not only on human health but also on the ecosystem are concerning, however the details of these effects are not known.
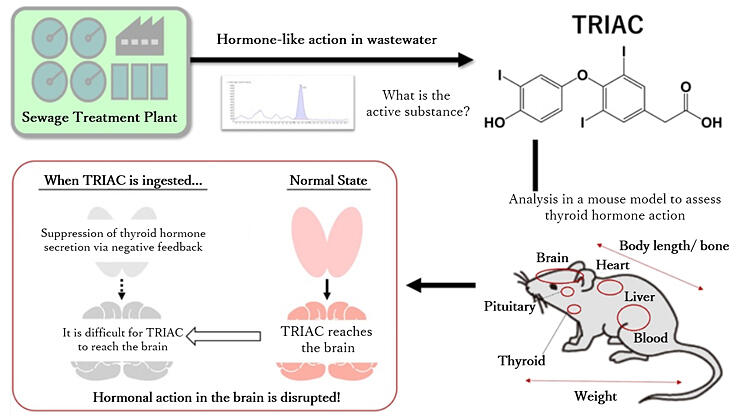
To evaluate the effects of TRIAC ingestion on the body, the research group developed a mouse model of hypothyroidism (hypothyroid mice). To establish this model, mice were given propylthiouracil (a drug used to treat Basedow's disease) to increase their sensitivity to thyroid hormones. In the experiment, the hypothyroid mice were given either TRIAC or T3, and the effects of the drugs in the brain, liver, and other major organs involved with thyroid hormones were compared.
The results showed that TRIAC was unable to restore the symptoms of thyroid hormone deficiency in the brain only. To clarify the cause, the amount of TRIAC in each organ of the TRIAC-treated mice was measured, whereupon it was found that this compound is not easily transported to the brain. It appears that TRIAC, in contrast to T3, has difficulty in passing through the blood-brain barrier.
On the basis of these results, T3 and TRIAC were respectively administered to normal mice, and the effects were analyzed. The body of the TRIAC-treated mouse sensed the presence of the thyroid hormone and initiated a negative feedback response to decrease endogenous T3 production, resulting in a decreased blood T3 level. It has been confirmed that thyroid hormone function in the brain is reduced because it does not reach this organ.
Although TRIAC is sometimes used overseas for the treatment of monocarboxylate transporter 8 (MCT8) deficiency, a hereditary deficiency of the T3 transporter, it has been found that its administration can decrease T3 in the brain and actually worsen the condition.
Yamauchi said, "In the future, we would like to conduct environmental research on TRIAC that has not yet progressed, such as where it exists in the environment and how it affects diseases and health."
Journal Information
Publication: iScience
Title: TRIAC disrupts cerebral thyroid hormone action via negative feedback and heterogenous distribution among organs
DOI: 10.1016/j.isci.2023.107135
This article has been translated by JST with permission from The Science News Ltd. (https://sci-news.co.jp/). Unauthorized reproduction of the article and photographs is prohibited.




