Infertility is a health problem faced by 15% of couples worldwide. In vitro fertilization/embryo transfer is one of the methods used to treat infertility. However, only ∼30% of the treatment succeed even if good quality of embryos are transferred into uteri, due to implantation failures. To address this issue, a research group led by Professor Yasushi Hirota and specially appointed researcher Shizu Aikawa from the Department of Obstetrics and Gynecology at the University of Tokyo Hospital, used human endometria and genetically modified mice, demonstrating that epigenomic abnormalities in endometria can cause implantation failures.
The research group first searched for genes that are differentially expressed in implantation-stage human endometrium between groups with successful pregnancy or recurrent implantation failures (RIFs). They found that highly expressed genes in endometria with RIFs were the targeted by trimethylation on lysine27 of histone H3(H3K27me3), a major epigenomic modification that represses gene expressions. Additionally, they found that RIFs group showed the reduced level of enhancer of zeste homolog 2 (EZH2), a key enzyme involved in the H3K27me3 modification.
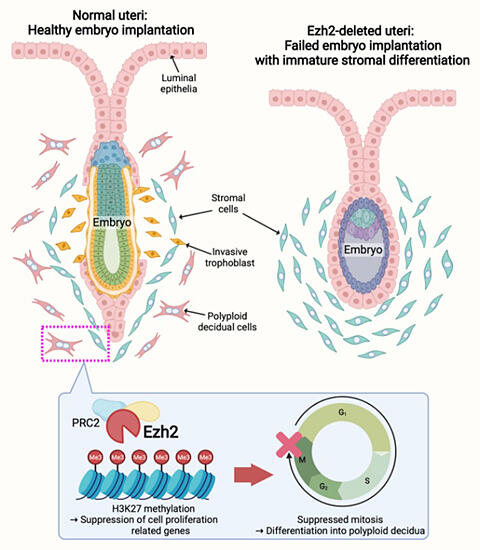
Next, they analyzed mice with uterine-specific deletion of EZH2 and found the expressions of cell division (mitosis)-related genes were reduced in this endometria during the implantation stage. In the normal endometrium, uterine stromal cells differentiate to the ones with polyploid features called decidual cells, which promotes embryo implantation and the following placentation. However, in the EZH2-deficient uteri, this stromal differentiation occurred poorly, eventually giving rise to flawed implantation.
In summary, this study revealed that repressive histone modification by EZH2 safeguards embryo implantation. In the future, evaluation of uterine cell differentiation might be applied for the development of new methods for diagnosing and treating implantation failures.
Regulation of implantation by EZH2
Journal Information
Publication: Cell Death & Disease
Title: The EZH2−PRC2−H3K27me3 axis governs the endometrial cell cycle and differentiation for blastocyst invasion
DOI: 10.1038/s41419-023-05832-x




