Catalysts alloyed with platinum (Pt) and transition metals such as cobalt (Co) have practical use as fuel-cell catalysts. However, although these materials exhibit higher activity than Pt, they have durability issues. In response to this issue, a research group led by Professor Toshimasa Wadayama and Graduate Student Yoshihiro Chida of the Graduate School of Environmental Studies at Tohoku University, has extensively investigated Pt-based high-entropy alloys (Pt-HEAs) composed of an HEA containing a mixture of five elemental species alloyed with Pt as a material that combines high activity with excellent durability.
The group recently fabricated model catalysts with controlled alloy top-surface structures on which the catalytic reaction proceeds and constituent-element distributions and revealed that the Pt-HEA surface has both high activity and durability. This achievement was published in Nature Communications.
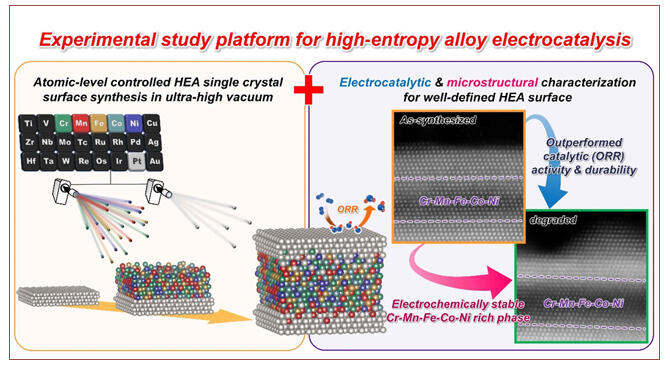
©Yoshihiro Chida et al.
The research group used arc plasma deposition to create model catalyst surfaces with an atomical level controlled near-surface constituent-element distribution and a top-surface structure composed of Pt and HEA to clarify the effect of surface microstructure on the activity and durability of the resulting catalyst for the oxygen reduction reaction (ORR). The team also established an experimental research platform for HEA electrocatalysts to clarify the mechanism of ORR property enhancement.
The relationship between the surface microstructures of the single-crystal surface atomic arrangements Pt (111, 110, 100) and HEAs containing Cr, Mn, Fe, Co, and Ni was evaluated using this platform, and the ORR properties of the catalysts were investigated. The experiments demonstrated that the Pt-HEA catalysts exhibited ORR properties superior to those observed in catalysts prepared from conventional Pt and Co alloys.
Specifically, the Pt(111)-HEA and Co alloys had similar surface activities. However, the Pt-HEA alloy demonstrated over 30% higher activity after the accelerated aging test. Furthermore, the initial ORR activity of the Pt(110)-HEA and Pt(100)-HEA catalysts was significantly higher than that of the Co alloy. Moreover, the Pt-HEAs maintained 2−3 times higher activity than the Co alloy even after aging.
The results showed that increasing both the number of alloying elements and the entropy of the alloy can effectively enhance the activity and durability of Pt-alloy catalysts.
This research was undertaken with funding from KAKENHI and NEDO.
Journal Information
Publication: Nature Communications
Title: Experimental study platform for electrocatalysis of atomic-level controlled high entropy alloy surfaces
DOI: 10.1038/s41467-023-40246-5
This article has been translated by JST with permission from The Science News Ltd. (https://sci-news.co.jp/). Unauthorized reproduction of the article and photographs is prohibited.




