A research group led by Professor Hitoshi Okazawa and Part-time Lecturer Kyota Fujita of the Medical Research Institute at Tokyo Medical and Dental University (TMDU) collaborated with the Tokyo Metropolitan Geriatric Hospital and Institute of Gerontology to investigate the propagation mode of α-synuclein, a protein responsible for Parkinson's disease, and have discovered a new mechanism for its transmission. The researchers analyzed the diffusion of α-synuclein expressed in mouse brains. Their findings revealed that α-synuclein was quickly transported to distant sites in a monomeric state via the intracerebral lymphatic system to form aggregates at the destination. The same mechanism likely also exists in neurodegenerative diseases associated with abnormal proteins, meaning that it is expected to become a new therapeutic target for these diseases. The results were published in the August 16, 2023 issue of the international academic journal Cell Reports.
Provided by TMDU
Parkinson's disease is a typical neurodegenerative disorder affecting approximately 200,000 patients in Japan, a number that continues to rise. The condition is characterized by the deposition and aggregation of α-synuclein in the brain neurons. α-Synuclein is transmitted from neurons in the intestinal tract to the brain stem via the vagus nerve. The abnormal protein is purportedly transmitted and propagates like a virus (prion-like hypothesis); a similar pathology is thought to occur in other conditions where aggregation has been observed, such as Alzheimer's disease. α-Synuclein aggregates were previously reported in fetal neurons transplanted to patients with Parkinson's disease. Lewy bodies (protein masses) were also observed when α-synuclein aggregates were injected into mouse brains, resulting in Parkinson's disease-like symptoms.
The research group used mice expressing α-synuclein for a long time in the lateral orbital area projecting from the olfactory bulbーa pathway where α-synuclein is thought to be transmittedーto investigate how α-synuclein spreads in the brain. From this they assessed the spread of α-synuclein throughout the brain two weeks after administration. They detected a low-molecular-weight, monomeric protein in neuronal regions distant from the injection site. The researchers also confirmed that the particles had transformed into aggregates at the destination after 12 months.
Previously, the predominant hypothesis was that α-synuclein was first formed in aggregates, which were then transmitted from neuron to neuron. However, the α-synuclein that propagated far into the brain was a monomer, which is inconsistent with this hypothesis.
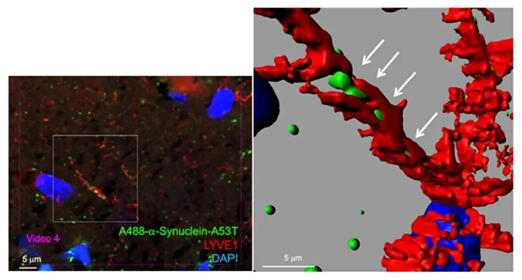
In response to this, the researchers investigated the positional relationship between vascular endothelial cells and monomers from multiple directions using a super-resolution microscope. They detected no monomers within the blood vessels. Next, they investigated the positional relationship between lymphatic vessels and epithelial cells and confirmed the presence of α-synuclein within lymphatic vessels.
Lymphatic vessels branch from thick lymphatic vessels along the external surfaces of blood vessels and finally reach the neurons, where they form aggregates. Since the monomers that cause Parkinson's disease are rapidly transported throughout the brain, the disease may progress more quickly than previously assumed. The research group clarified that the propagation mode did not contradict the conventional concept; rather, these findings revealed a new propagation route, lymphatic vessels.
Until now, therapeutic drugs for neurodegenerative diseases have targeted aggregates; however, the current study's findings suggest that targeting smaller monomers and oligomers may lead to the development of more effective therapeutic drugs. However, much remains unknown regarding the role of the lymphatic system in the brain.
Okazawa stated, "Research on lymphatic vessels is still in its early stages, and I hope to see how the results will be received by researchers in this field. Concurrently, I would like to continue our research on ways to mitigate the damage caused by toxic products of intracellular and extracellular origin in neurodegenerative diseases."
This article has been translated by JST with permission from The Science News Ltd. (https://sci-news.co.jp/). Unauthorized reproduction of the article and photographs is prohibited.




