Ongoing studies have aimed to provide radical treatment for regenerating neurons lost due to stroke by converting existing cells to neurons rather than inserting cells in the brain. However, thus far, no study has successfully developed such treatments. A research group led by Dr. Takashi Irie of Kyushu University Hospital and Lecturer Taito Matsuda, Professor Noriko Isobe, and Professor Kinichi Nakashima of the Graduate School of Medical Sciences at Kyushu University have successfully restored neurological function in mice with brain injury by differentiating microglia, which aggregate at the site of brain injury, directly into neurons via direct reprogramming. "Although many challenges still exist, including developing an efficient system for direct reprogramming of human cells, we hope that the results of our ongoing experiments will contribute to clinical practice," stated Nakashima. The study results were published in PNAS.
Provided by Kyushu University
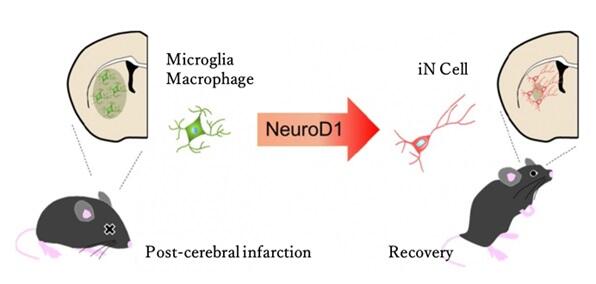
Before the current study by the research group, while an approach to directly convert astrocytes into neurons had been reported, no studies had reported the successful recovery of neurological function because of the insufficient number of neurons produced by that approach. Therefore, the research group targeted the transdifferentiation of microglia aggregating at the site of brain injury to neurons, aiming to potentially improve functional damage without depleting the source cells.
In a previous study, they had demonstrated that in mice, microglia could be converted to neurons by introducing the transcription factor NeuroD1 (ND1). In the current study, an adult mouse model of focal stroke was generated to investigate the direct conversion of microglia to neurons and its effects. Seven days after stroke was induced in the mouse model, microglia aggregation was observed in the striatum at the core of the lesion where neuronal loss was prominent. Adding a lentivirus that expresses ND1 in microglia to the stroke lesion induced the differentiation of the microglia into neurons. Over eight weeks after stroke, newly generated neurons were replenished, and the area of the neuronal loss decreased notably compared with that in the untreated group.
In the ND1-expressing mice, the results of the tail suspension test started to improve two weeks after brain injury and recovered to a level similar to that in normal mice at eight weeks. In other words, the researchers observed the effects of the functional integration of the newly generated neurons into the existing neuronal circuits. However, the recovered function was lost when the neurons derived from microglia were removed from functionally recovered mice.
The results of the study demonstrated the possibility of functional recovery after stroke through the direct contributions of neurons newly generated through direct reprogramming. Because the approach in the current study used microglia that aggregate at the site of brain injury, this method can not only restore physically injured brain sites but may also have the potential to newly generate neurons lost during Alzheimer's Disease and other conditions.
Irie commented, "As a neurologist, I see limitation in treatment in the current clinical practice. I hope that our findings will be translated into clinical practice in the future."
Matsuda stated, "Considering the clinical applications of our findings, we wanted to publish our article sooner, but it took four years to publish it after our first submission. This is a good approach, so we hope to utilize it for a broad range of applications."
Journal Information
Publication: PNAS
Title: Direct neuronal conversion of microglia/macrophages reinstates neurological function after stroke
DOI: 10.1073/pnas.2307972120
This article has been translated by JST with permission from The Science News Ltd. (https://sci-news.co.jp/). Unauthorized reproduction of the article and photographs is prohibited.




