A research group led by Associate Professor Yasuhiro Shiraishi, Graduate Student Masahiro Jio, and Professor Takayuki Hirai of the Division of Chemical Engineering at the Department of Materials Engineering Science/Research Center for Solar Energy Chemistry of the Graduate School of Engineering Science at Osaka University has announced the development of a Nafion (Nf)-containing resorcinol-formaldehyde (RF@Nf) photocatalytic resin that shows considerably high hydrogen peroxide (H2O2) generation ability under solar irradiation using water and oxygen (O2) as raw materials. H2O2 has attracted attention as an energy carrier because it can be used as a fuel for fuel-cell power generation. It is also an important chemical used as a bleaching agent and disinfectant and has promising potential applications for realizing a hydrogen energy society. The results were published in the July 24, 2023 issue of the international academic journal JACS Au.
Provided by Osaka University
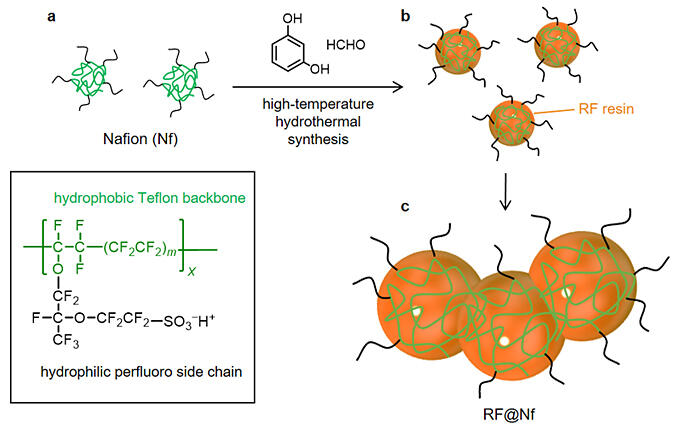
In this study, the properties of RF resin were improved using Nafion (Nf), a general-purpose synthetic polymer. Nf is an amphiphilic polymer composed of a hydrophobic Teflon backbone and hydrophilic perfluorinated side chains with sulfonic acid groups. It is widely used as an ion-exchange membrane in fuel cells. When resorcinol, formaldehyde, and an Nf dispersion are added to water and subjected to high-temperature water treatment (∼250 ℃), the hydrophobic backbone of Nf is incorporated into the RF resin while its hydrophilic side chains face outward. Therefore, the growth of the resin is suppressed via electrostatic repulsion owing to the hydrophilic groups of Nf.
RF resin is made up of spherical particles with a diameter of ∼3 µm, whereas RF@Nf has a consolidated structure consisting of small particles with a diameter of ∼0.3 µm. When RF resin is suspended in water and irradiated with light in the presence of O2, H2O2 is formed via water oxidation and O2 reduction. However, it is also decomposed by these reactions.
In the case of RF@Nf, the specific surface area of the resin is increased by the reduction in its particle size, which promotes H2O2 generation, and the resin surface is made hydrophobic by the incorporation of the hydrophobic portion of Nf. This means that the generated H2O2 is prevented from approaching the resin surface and decomposing.
Compared with RF resin, RF@Nf resin can enable the synthesis of up to 1.5 times more H2O2 solution. This concentration is the highest amount of H2O2 generated using powdered photocatalysts reported to date. Metal-free catalysts synthesized using general-purpose ion-exchange polymers were found to be effective for artificial photosynthetic H2O2 production. This strategy enables on-demand synthesis, in which H2O2 solution can be easily synthesized in the desired amount at the desired location.
Shiraishi said, "Using the catalyst we have developed, it will be possible to produce H2O2 solution from water and air under normal temperature and pressure conditions at low costs utilizing solar energy. Although several issues, such as the formation of an insufficient amount of H2O2, remain to be resolved, we are hopeful that we have made a step forward toward a hydrogen energy society."
Journal Information
Publication: JACS Au
Title: Nafion-Integrated Resorcinol-Formaldehyde Resin Photocatalysts for Solar Hydrogen Peroxide Production
DOI: 10.1021/jacsau.3c00262
This article has been translated by JST with permission from The Science News Ltd. (https://sci-news.co.jp/). Unauthorized reproduction of the article and photographs is prohibited.




