To realize a hydrogen society, materials for producing, storing, transporting and preserving hydrogen are essential. However, because understanding the behavior of hydrogen in materials requires special equipment and environments, the scope of observation has been extremely limited.
Assistant Professor Hiroshi Kakinuma and his colleagues at the Institute of Materials Research at Tohoku University, have developed a simple and cost-effective method to capture moving images of hydrogen flowing through metals. This new technology uses polyaniline, a polymer that changes color when it reacts with hydrogen atoms, and a general-purpose optical microscope. The results of this study were published in the journal Acta Materialia.
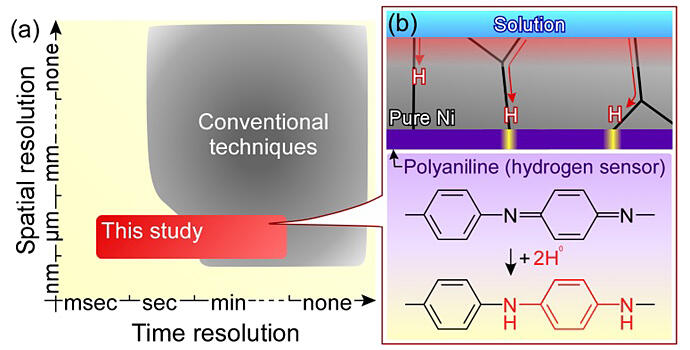
(b) Schematic of the present hydrogen visualization technique.
Provided by the Institute for Materials Research at Tohoku University
Polyaniline undergoes a change in color upon reacting with hydrogen atoms in metals, enabling the visualization of hydrogen distribution within metals. An optical microscope can capture videos in micro-scale spatial resolution across a wide sub-millimeter field of view. Observing the color changes of polyaniline using an optical microscope enables observation of the behavior of hydrogen within a wide field of view with high spatial and temporal resolutions, far surpassing conventional technologies.
In this study, the hydrogen flow through a pure nickel foil was observed. The diffusion of hydrogen in a pure nickel foil is driven by concentration gradients. Eventually, hydrogen reaches the opposite side of the pure nickel foil, which is coated with polyaniline. Polyaniline reacts with hydrogen atoms reaching it through the metal foil, changing color from purple to white. By observing this change in color using an optical microscope, the behavior of hydrogen in pure nickel could be analyzed. This analysis revealed that hydrogen preferentially diffused through the crystal grain boundaries, where the arrangement of nickel atoms was disturbed.
It was revealed that this hydrogen diffusion varies depending on the grain boundary type in the crystal. This suggests that the diffusion of hydrogen atoms in pure nickel depends on the arrangement of nickel atoms, indicating that larger geometric spaces in grain boundaries result in a greater hydrogen flow.
The relationship between the atomic-level structural features of metallic materials and the behavior of hydrogen was experimentally elucidated. This was achieved for the first time using hydrogen observation technologies with high spatial and temporal resolution, which cannot be achieved using conventional methods.
The newly developed hydrogen observation method can be applied to any metal. This method is expected to experimentally elucidate the relationship between the structures of various metals and the behavior of hydrogen, which was discussed in previous studies only through simulations. Clarifying the causal relationship between atomic-level metal structure and microscale hydrogen behavior will enable efficient material design.
This article has been translated by JST with permission from The Science News Ltd. (https://sci-news.co.jp/). Unauthorized reproduction of the article and photographs is prohibited.




