Upper tract urothelial carcinoma (UTUC) has a high recurrence rate and so postoperative follow-up is currently performed using invasive methods or tests with low sensitivity. A research group led by Graduate Student Daichi Tamura and Professor Wataru Obara of the Department of Urology at Iwate Medical University and Professor Satoshi Nishizuka of the Division of Biomedical Research and Development at Iwate Medical University Institute for Biomedical Sciences, in collaboration with Professor Yutaka Suzuki of the Department of Computational Biology and Medical Sciences at the Graduate School of Frontier Sciences at the University of Tokyo demonstrated that postoperative UTUC recurrence can be detected by conducting a follow-up test for patient-specific cancer-derived DNA in blood and urine. The study was published in Cancer Science.
Provided by Iwate Medical University
The research group focused on 23 patients who received surgery for UTUC. DNAs were extracted from tumor tissues obtained from surgical specimens and used for both whole-exome sequencing through next-generation sequencing (NGS) and TERT promoter sequencing.
Plasma and urine samples were first collected before and after surgery, and the sample supernatants (the liquid obtained after centrifugation of the sample) were then obtained for DNA extraction. Based on the results of gene mutation analysis of the tumor tissues, patient- specific gene mutations were selected for each patient for follow-up, and the variant allele frequencies (VAFs) were monitored for two years after surgery using the digital polymerase chain reaction (dPCR).
The primer/probe sets used for the dPCR analysis were selected from the library mainly developed by Iwate Medical University. The aims of the study were to compare the effectiveness of monitoring postoperative circulating tumor DNA (ctDNA) as a biomarker for UTUC recurrence against existing testing methods (computed tomography (CT), cystoscopy, and urine cytology) and to examine whether the VAF and its changes in ctDNA are associated with postoperative recurrence.
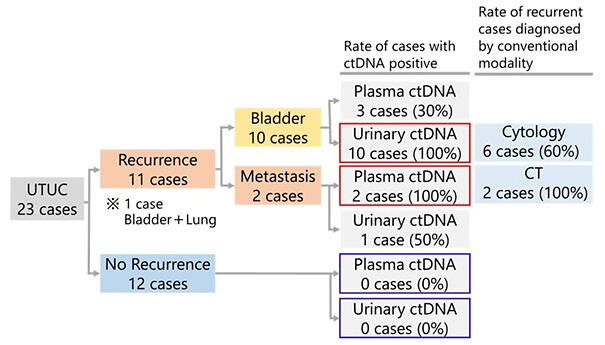
As a result of selecting patient-specific gene mutations for each patient for monitoring, 83% of the study participants were found to have mutations listed in the OTS1000ex library. By designing novel probes for the remaining patients, monitoring of ctDNA was possible for all the study participants. The dPCR analysis of the preoperative plasma and urine samples detected patient-specific gene mutations in 96% of the patients.
Among the patients with postoperative recurrence, all those with bladder cancer recurrence were positive for urinary ctDNA at the time of recurrence, demonstrating the higher sensitivity of this method in detecting recurrence than that achieved with urine cytology. All patients with metastatic recurrence had ctDNA-positive plasma readings. In particular, in the patients with bladder cancer recurrence, their urine samples showed ctDNA-positive results approximately 2 months earlier than with the conventional testing methods. Among the patients without postoperative recurrence, all remained ctDNA negative three months after the surgery, when the conventional follow-up was initiated, and thereafter.
The rates of decrease in VAF from pre- to post-surgery between the patients with postoperative recurrence and those without were compared. The comparison revealed that the rates of decrease in urinary VAFs from before the surgery to two days thereafter was significantly lower in the recurrent cases. In the analysis of risk factors, the recurrence-free survival period was significantly shorter in patients with a lower rate of decrease in urinary ctDNA VAFs at postoperative day 2.
These findings suggest that ctDNA testing may predict the risk of postoperative recurrence and may be useful in determining therapeutic indications for adjuvant therapy. Currently, blood ctDNA monitoring for cancer is available at Iwate Medical University at the patient's own expense.
Journal Information
Publication: Cancer Science
Title: Postoperative recurrence detection using individualized circulating tumor DNA in upper tract urothelial carcinoma
DOI: 10.1111/cas.16025
This article has been translated by JST with permission from The Science News Ltd. (https://sci-news.co.jp/). Unauthorized reproduction of the article and photographs is prohibited.




