A research group led by Professor Shingo Hatoya and Visiting Researcher Masaya Tsukamoto (also a researcher at the National Center for Child Health and Development) of the Graduate School of Veterinary Science at Osaka Metropolitan University, which has been collaborating with Anicom Specialty Medical Institute Inc., and TOKIWA-Bio Inc., announced that they have achieved the stable production of canine induced pluripotent stem (iPS)cells. By identifying six reprogramming genes and introducing them into cells derived from the urine of dogs, the efficiency of iPS cell production was improved by approximately 120 times compared with that of the conventional method using fibroblasts. The research group further succeeded in producing canine iPS cells without using feeder cells.
In the future, the group plans to provide canine iPS cells to researchers throughout the world, aiming to realize regenerative medicine in veterinary medical care. The achievement is expected to lead to the further development of regenerative therapy in veterinary medicine. The study results were published in the December 22 issue of the international academic journal Stem Cell Reports.
Provided by Shingo Hatoya, Osaka Metropolitan University
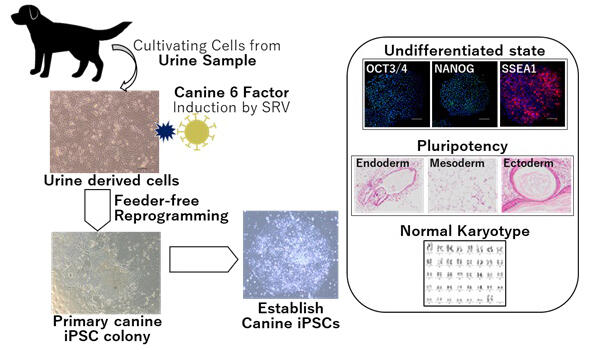
iPS cells are already being used for regenerative therapy in humans and are expected to be applied in veterinary medicine as well. Prior to this study, the research group had been working to create canine iPS cells by introducing four human reprogramming genes into fibroblasts (a precursor cell of connective tissue), but the problem was the low production efficiency. Because identification of the estrous cycle in dogs is challenging, obtaining fertilized canine eggs for research is difficult. The lack of knowledge and research progress in canine embryonic stem cells had compounded the problem.
The research group then considered six genes (LIN28A, OCT3/4, SOX2, C-MYC, KLF4, NANOG) instead of the four human reprogramming genes that had been so far used. Furthermore, they considered introducing these genes into urine-derived cells, which are less stressful to collect.
A comparison of the canine and human sequences of these six genes revealed that only NANOG had a low similarity between the two species. Therefore, the six canine factors were selected. Two types of virus vectors with different gene expression levels were created by loading the six canine factors onto Sendai virus vectors. Each of the two types of vectors was introduced into fibroblasts, which were then cultured on feeder cells (a human iPS cell medium).
The reprogramming efficiency was improved by approximately 9 times compared with that of conventional methods. It was confirmed that the canine reprogramming gene promoted the reprogramming of canine somatic cells.
The generated canine iPS cells were treated with small interfering RNA (siRNA) to remove the vector, and their properties were assessed. These cells were confirmed to maintain an undifferentiated state as pluripotent stem cells, retaining the ability to differentiate into three germ layers. It was also confirmed that iPS cells can be produced using adult dog-derived fibroblasts, although the production efficiency was lower. Furthermore, the research group found that canine iPS cells could be generated even by culturing canine fetal fibroblasts under feeder-free conditions.
Finally, when the six canine factors were introduced into canine urine-derived cells by the vectors, iPS cells were successfully generated. The production efficiency was 0.15−2.43%, approximately 120 times more efficient than conventional methods.
As a pluripotent stem cell, the canine iPS cells maintained an undifferentiated state and had the ability to differentiate into three germ layers, and a karyotypic analysis confirmed no abnormalities at the chromosomal level. Fabrication was possible even without a feeder, although the efficiency was low at 0.05%.
Urine samples from five beagle dogs were used in the study, and the production efficiency differed depending on the individual and on the gene expression level of the vector.
In the future, the research group will share the established iPS cells widely with researchers to promote iPS cell research in veterinary care. This technology will extend to applications in treating animal diseases, elucidating disease mechanisms, and screening in drug discovery. The laboratory group is currently investigating the possibility of producing blood products by differentiating the cells into red blood cells.
Hatoya said, "Compared with medical research for humans, veterinary research on dogs is lacking in personnel and research funds. Even under these circumstances, and after much trial and error, we have now been able to generate canine iPS cells efficiently and safely. As a veterinarian, I have examined many animals, yet many diseases exist that remain untreatable or are not well understood. In the future, I aim to conduct research to create various cells in the body using canine iPS cells and apply the results to treat sick dogs and elucidate various pathological conditions."
Journal Information
Publication: Stem Cell Reports
Title: Generation of canine induced pluripotent stem cells under feeder-free conditions using Sendai virus vector encoding six canine reprogramming factors
DOI: 10.1016/j.stemcr.2023.11.010
This article has been translated by JST with permission from The Science News Ltd. (https://sci-news.co.jp/). Unauthorized reproduction of the article and photographs is prohibited.




