A research group led by Professor Koji Aoki of the Faculty of Medicine at the University of Fukui announced on February 6 that they have clarified the mechanism of colorectal cancer initiation. They demonstrated that mutations of causative genes led to an increase in β-catenin proteins and activated RNA polymerase II, which further induced the expression of cancer stemness-related genes. Additionally, the inhibition of cyclin-dependent kinase 12 (CDK12), a downstream molecule of the polymerase-associated factor 1 (PAF1) complex that activates the aforementioned enzyme, suppressed colorectal cancer stemness and tumor growth in mice. The findings are expected to lead to the development of therapeutic drugs for colorectal cancer. The study results were published in the international academic journal Oncogene.
Provided by the University of Fukui
Mutations in the adenomatous polyposis coli (APC) gene are observed in 50−80% of patients with colorectal cancer. Benign tumors derived from the aforementioned mutations turn into malignant tumors (cancerization). The APC mutations cause an increase in β-catenin proteins and induce the expression of genes that promote cancer stemness. The mechanism of this gene expression induction had been unclear.
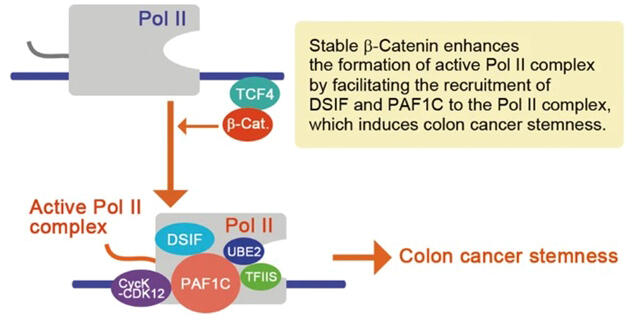
Previously, the research group demonstrated that the inhibition of β-catenin proteins in colorectal cancer cells induced changes in the dynamics of RNA polymerase II. This result suggested that the PAF1 complex, which binds to and enhances the transcription of the aforementioned enzyme, was possibly impaired. The PAF1 complex consists of six proteins, including PAF1, which is a main component.
In the present study, the research group investigated the role of the PAF1 gene in colorectal cancer cells. First, the researchers generated colorectal cancer cell lines in which the PAF1 gene was artificially suppressed, which resulted in a decrease in the expression of various genes that could be markers of colorectal cancer stemness (including LGR5). The results revealed that PAF1 proteins are essential for the expression of the aforementioned colorectal cancer stemness-related genes. The research group also confirmed that tumor growth was suppressed in mice that had been subcutaneously transplanted with colorectal cancer cells in which PAF1 gene expression was inhibited.
Next, to assess the relationship between β-catenin and the PAF1 complex, the process through which RNA polymerase II forms a complex to function (active RNA polymerase II—PAF1 complex) was investigated. Because this complex is formed in stages, the research group tagged and then expressed each component factor of the complex in a single cell and examined the stage in which its formation state is changed by β-catenin. The results showed that β-catenin enhanced formation of the complex. Furthermore, suppression of CDK12, a factor necessary for the polymerase II complex to function, led to the reduced expression of genes related to colorectal cancer stemness. Tumor growth was confirmed to be suppressed in colorectal cancer cells that were cultured with CDK12 inhibitors, even after their subcutaneous transplantation into mice.
Journal Information
Publication: Oncogene
Title: NELF and PAF1C complexes are core transcriptional machineries controlling colon cancer stemness
DOI: 10.1038/s41388-023-02930-0
This article has been translated by JST with permission from The Science News Ltd. (https://sci-news.co.jp/). Unauthorized reproduction of the article and photographs is prohibited.




