A research group led by Lecturer Yuji Atsuta of the Faculty of Science at Kyushu University (former researcher at Harvard Medical School, USA) and Professor Clifford J. Tabin of the Department of Genetics at Harvard Medical School announced that they have identified a group of reprogramming factors that can impart the properties of limb-producing limb progenitor cell (LPC) to the lateral plate mesoderm using mouse cells. Additionally, they have successfully established a reprogramming method that generates LPCs. The results are expected to lead to the realization of limb regenerative medicine. The study results were published in the February 5, 2024 issue of the international academic journal Developmental Cell.
Provided by Kyushu University
The major tissues of the hands and feet (limbs) are formed during the fetal (embryonic) stage and are derived from a mesodermal tissue called the lateral plate mesoderm. The formation of the limb bud, the source of the limb, is initiated by the specification of some lateral plate mesodermal cells into a population of LPCs. As development progresses, the LPCs differentiate into connective tissues such as bone, cartilage, ligaments, tendons, and dermis within the limb. Aside from forming the limbs, the lateral plate mesoderm also gives rise to other organs and tissues, such as the heart and body cavity walls.
Limb buds consist mainly of LPCs and the epidermal cells that cover them, and many genes involved in limb bud formation and limb bud growth factors have been identified. However, the intracellular factors that initially specify LPCs in the lateral plate mesoderm and differentiate them from other lateral plate mesoderm-derived tissues were unknown. Therefore, the aim of this study was to identify reprogramming factors among the genes expressed in limb buds that can convert cells of non-limb origin into LPC-like cells.
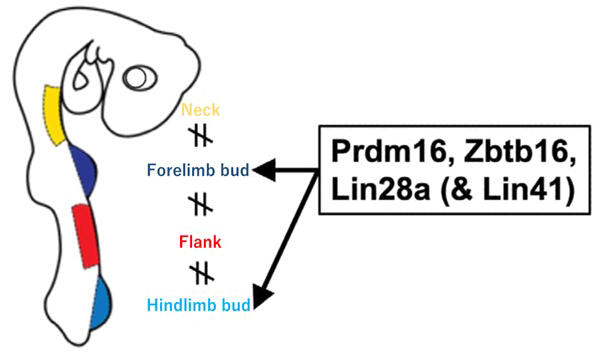
The researchers hypothesized that factors that are specifically expressed in the planned limb region and that can impart LPC properties to non-LPC cells were most likely to also play a role in specifying LPCs during limb development. RNA sequencing was used to comprehensively search for genes that are highly expressed in limb buds and poorly expressed in the lateral plate mesoderm region of the neck and flanks. As a result, 18 candidate genes were identified.
The reprogramming factors were narrowed down using a drop-out assay in which one gene each was omitted from the transfection experiment. As a result, transfection of the transcription factor Prdm16, the transcription factor Zbtb16, and the RNA-binding factor Lin28a (PZL) was found to be sufficient to express the LPC marker genes.
Additionally, the expression of the E3 ubiquitin ligase Lin41 was shown to be upregulated in the early stages of successful reprogramming in the cells. Therefore, when Lin41 was introduced into fibroblasts in combination with PZL (PZLL combination), the reprogramming efficiency was increased. In the future, knockout mice will be generated to test whether the identified reprogramming factors are actually necessary for LPC identification.
Atsuta said, "We started this research in the USA approximately nine years ago, and with the cooperation of various people, we were finally able to find genes that impart the properties of limb-derived cells (limb progenitor cells) to non-limb-derived cells. Our goal is to further develop this research to establish a reprogramming method to create human limb progenitor cells and produce results that will contribute to the development of regenerative medicine."
Journal Information
Publication: Developmental Cell
Title: Direct reprogramming of non-limb fibroblasts to cells with properties of limb progenitors
DOI: 10.1016/j.devcel.2023.12.010
This article has been translated by JST with permission from The Science News Ltd. (https://sci-news.co.jp/). Unauthorized reproduction of the article and photographs is prohibited.




