A research group led by Professor Kei-ichiro Ishiguro and Assistant Professor Ryuki Shimada of the Institute of Molecular Embryology and Genetics and Graduate Student Saori Yoshimura of the Graduate School of Medical Sciences at Kumamoto University, in collaboration with the Institute of Advanced Medical Sciences at Tokushima University, has announced that they identified "HSF5," a gene that controls meiosis progression in spermatogenesis. The HSF5 protein is classified as a heat shock factor. Surprisingly, they have discovered that HSF5 is not involved in the heat shock response but is responsible for the regulation of the meiosis completion process and spermatogenesis. They also confirmed that the HSF5 gene disorder caused infertility due to spermatogenic failure. The results are expected to contribute to the elucidation of the cause of infertility with spermatogenic failure and were published in the April 29 issue of the international academic journal Nature Communications.
Provided by Kumamoto University
Male infertility accounts for approximately half of infertility issues, and 70%−90% of male infertility is attributed to spermatogenic failure. However, the causes of spermatogenic failure in approximately 40% of cases are unknown. In the ovary and testis, eggs and sperm are formed through meiosis, respectively, which is different from the process of somatic cell division.
In 2020, the research group discovered MEIOSIN, a gene that directs initiation of meiosis. The MEIOSIN protein was shown to bind to and simultaneously activate approximately 400 genes involved in meiosis. In this study, the research group analyzed "HSF5," a gene that binds to and is activated by MEIOSIN and has unknown functions. The HSF5 protein is classified as a DNA-binding protein called heat shock factor (HSF) based on its structure. While HSF1 to HSF5 paralogs are known, the function was unknown only for HSF5.
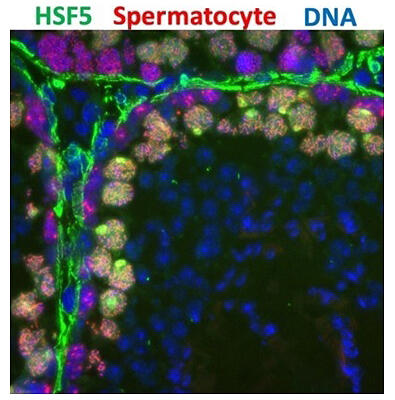
HSFs 1 through 4 are known to protect cells against heat. First, they used the polymerase chain reaction to identify organs expressing HSF5 and found that it was specifically expressed in the testis. It was found to function in the nucleus of spermatocytes from around the middle to the end of meiosis. Next, to investigate the function of HSF5, they disrupted the HSF5 gene in mice via genome editing. The results showed that in the testes of the genome-edited mice, spermatocytes started meiosis, but died during the process, causing male infertility. The testes of these mice were underdeveloped and atrophied.
To further investigate the molecular function of HSF5, they analyzed big data obtained by the ChiP-seq method. HSF5 was found to bind to the promoter regions of about 200 genes involved in the spermatogenesis process and activate these genes. The spermatogenesis process in which HSF5 was shown to be involved here is completely different from the heat response, in which HSF5 was initially thought to be involved because spermatogenesis is susceptible to heat. Furthermore, they examined the testicular cell population from normal and HSF5-deficient mice by single-cell RNA-seq and confirmed that the cell population preparing for spermatogenesis was lost in the HSF5-deficient mice. These findings indicate that HSF5 binds to a number of genes, ensures meiosis completion, and simultaneously activates the spermatogenesis program.
Ishiguro commented, "Mice were used in our experiments, but the HSF5 gene is also present in the human genome. The findings of this study are expected to contribute to the clarification of the pathophysiology of male infertility, particularly azoospermia, which presents with spermatogenic failure. Furthermore, we have identified numerous other genes regulated by MEIOSIN, and we hope to contribute further to the understanding of the causes of infertility by elucidating the functions of these genes in the future."
Journal Information
Publication: Nature Communications
Title: Atypical heat shock transcription factor HSF5 is critical for male meiotic prophase under non-stress conditions
DOI: 10.1038/s41467-024-47601-0
This article has been translated by JST with permission from The Science News Ltd. (https://sci-news.co.jp/). Unauthorized reproduction of the article and photographs is prohibited.




