In recent years, technologies for capturing carbon dioxide (CO2), a greenhouse gas, and effectively utilizing it in the production of biofuels and concrete products are being developed. Moreover, if the function of materials including CO2 can be controlled, new possibilities for its utilization will open up.
A research group led by Professor Yohei Miwa of the Gifu University Faculty of Engineering has developed a silicone elastomer that responds to certain gases including CO2, stretches satisfactorily, and resists tearing. Elastomers are a polymer material obtained from cross-linking polymer molecules. Because they exhibit rubber elasticity, they are widely used in our daily lives, such as in vehicle tires and rubber bands.
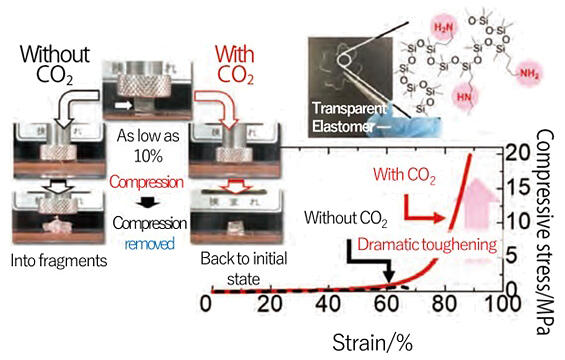
The elastomer developed by the group is easily synthesized by cross-linking polydimethylsiloxane with amines. In this elastomer, amines react quickly with CO2, whereupon the ionic components generate aggregate and act like a cushion, inhibiting fracture and making it tough. Thus, the elastomer can withstand high pressures of approximately 20 megapascals (MPa) (≈200 atm). 20 MPa is equivalent to the pressure of a light car on a one-yen coin.
This elastomer responds to the low concentrations of CO2 and acidic gases such as hydrogen chloride and acetic acid. Furthermore, this technology can be applied to other polymer materials such as polyethylene. Currently, the research group is developing a polymer material that hardens in response to CO2. In the future, they aim to synthesize a polymer material whose hardness and softness can be freely controlled by CO2 and to pioneer technologies for effective CO2 utilization.
Appearance of the developed silicone elastomer and toughening behavior in response to CO2.