A research group led by Professor Kazunobu Sawamoto of the National Institute for Physiological Sciences and Specially Appointed Assistant Professor Mami Matsumoto of the Institute of Brain Science, Graduate School of Medical Sciences at Nagoya City University has discovered that newly born nerve cells (new neurons) generated in the postnatal mouse brain migrate at high speed while a wide space between the cells is created during the collective migration. They have also found that when the brain is damaged by cerebral infarction, neuraminidase increases to reduce this space, impeding neuronal migration. Furthermore, administration of a neuraminidase inhibitor facilitated neuronal migration and promoted neuronal regeneration and functional recovery in injured brains in mice and primates. The work was published in EMBO Molecular Medicine.
Provided by Nagoya City University
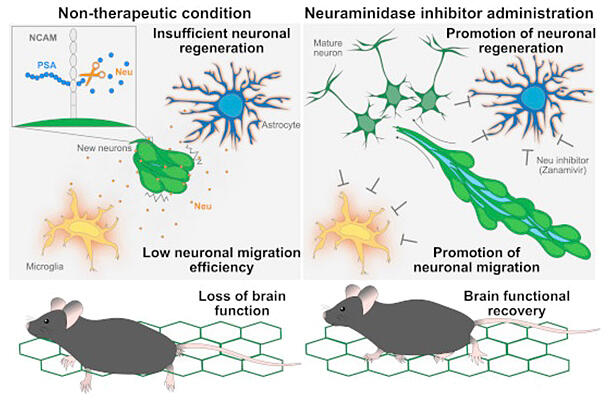
Stem cells that give rise to neurons are present in limited areas of the postnatal brain and during embryonic life. New neurons produced from neural stem cells form chains and migrate at high speed in the brain toward their destination, where they mature and are involved in the maintenance of brain function. Meanwhile, when brain injury occurs, some new neurons have been reported to migrate to the injured area and be involved in the regeneration of lost neurons. However, their migration efficiency is low and insufficient to restore brain functions lost because of injury.
The research group investigated why new neurons migrating in the normal brain can move at high speeds. Three-dimensional electron microscopic analysis revealed the presence of wide space between new neurons in chains. They found that this space is maintained by polysialic acid (PSA), whereby adhesion between new neurons is maintained in a proper state. In other words, new neurons migrating in the normal brain have an appropriate space in between by virtue of PSA and thereby move at high speed.
Next, three-dimensional electron microscopic analysis of new neurons migrating in the injured brain revealed reduced space and excessive cell adhesion. The measured levels of PSA, which is involved in space maintenance, were reduced in new neurons migrating in the injured brain. Furthermore, the expression of neuraminidase, which cleaves PSA, was elevated by brain injury. These results indicate that in new neurons migrating in the injured brain, brain injury induces a reduction in PSA levels by increasing neuraminidase, which cleaves PSA, resulting in reduced efficiency of neuronal migration due to excessive cell adhesion.
The researchers investigated whether inhibiting neuraminidase, which is increased during brain injury, could promote the migration of new neurons, efficiently supply them to the injured area, and contribute to the regeneration of brain function. Administration of the neuraminidase inhibitor Relenza, an anti-influenza drug, to cerebral infarction model mice suppressed PSA reduction and promoted the migration of new neurons to the injured area. Relenza treatment also resulted in a greater number of mature neurons in the injured area. The researchers also assessed the inhibitor's effects on impaired walking function, a common consequence of cerebral infarction, and observed improved walking function in mice treated with the inhibitor. Furthermore, they demonstrated in both mice and primates that Relenza treatment suppressed PSA reduction and promoted the migration of new neurons to the injured area.
These findings suggest that inhibiting the function of neuraminidase, which is increased by brain injury, can promote migration of new neurons, promote neuronal regeneration and restore brain function. The application of the mechanism revealed in this study is expected to contribute to the development of a new, minimally invasive and broadly effective treatment for brain diseases, for which radical treatment methods have not been established.
Journal Information
Publication: EMBO Molecular Medicine
Title: Neuraminidase inhibition promotes the collective migration of neurons and recovery of brain function
DOI: 10.1038/s44321-024-00073-7
This article has been translated by JST with permission from The Science News Ltd. (https://sci-news.co.jp/). Unauthorized reproduction of the article and photographs is prohibited.




