A joint research group led by Team Leader Yi-long Cui and Research Scientist (at the time of the study) Hiroyuki Neyama of the RIKEN Center for Biosystems Dynamics Research, Team Leader Yasuyoshi Watanabe of the Laboratory for Pathophysiological and Health Science of the RIKEN Center for Biosystems Dynamics Research, and Assistant Professor Yoshiki Miyasaka of the Institute of Experimental Animal Sciences, Graduate School of Medicine at Osaka University, has announced that they clarified the neurobiological basis of placebo analgesia, in which a sham drug (placebo) devoid of any pharmacological action actually relieves pain.
The research group focused on rat brain regions shown to be involved in the placebo effect by positron emission tomography (PET). It was found that "expectation" plays a role in alleviating pain. The findings are expected to contribute to the reduction of adverse drug reactions and prevention of dependence by reducing the dosage of analgesics. The results were published in the international journal Science Advances on January 15.
Provided by RIKEN
The placebo effect, in which a therapeutic effect is obtained simply by assuming that a placebo devoid of any pharmacological effect is effective, is well known and scientifically proven. However, its neurobiological basis has remained unknown, and its medical applications are limited. Recent studies using noninvasive functional brain imaging techniques such as functional magnetic resonance imaging (fMRI) and PET in humans have reported that placebo analgesia is associated with increased neuronal activity in brain regions, such as the dorsolateral prefrontal cortex, anterior cingulate cortex, and periaqueductal gray. It was also reported that endogenous opioids (bioactive substances with morphine-like activity) and the dopaminergic nervous system are involved in placebo analgesia.
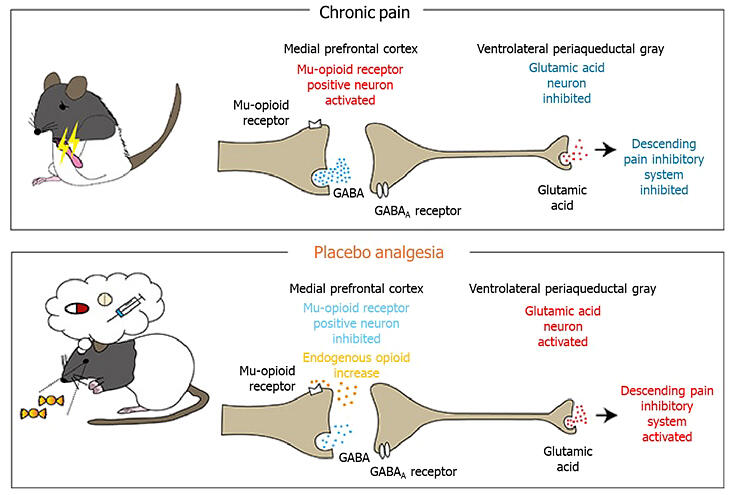
In this study, the research group developed genetically engineered rats in which the activity of MOR-positive neurons (neurons expressing mu-opioid receptors) in specific brain regions could be selectively manipulated and examined their effect on placebo analgesia. Previously, the research group used a rat model of neuropathic pain, which exhibits sensitivity to weak pain stimuli in the hindlimb, to demonstrate that pain is alleviated when saline is administered as a placebo following several days of conditioning with an analgesic. Therefore, they selectively stimulated the activity of neurons projecting from the medial prefrontal cortex (mPFC) to the ventrolateral periaqueductal gray (vlPAG) (projection neurons). As a result, placebo analgesia was blocked.
A pain stimulus signal is delivered to the spinal cord through sensory neurons, and this stimulus signal is transmitted from the spinal cord to the brain, where the pain is perceived. The brain's descending pain inhibitory system reduces this pain by suppressing the transmission of pain signals from the brain to the spinal cord.
Therefore, the group investigated which brain regions mediate the activation of the descending pain inhibitory system by MOR-positive neurons in the mPFC. They previously demonstrated that functional coupling between the mPFC and the vlPAG, a key region involved in pain control, is enhanced in response to placebo analgesia. Based on this, they investigated the relationship between the neuronal circuit connecting the mPFC to the vlPAG and placebo analgesia. The results showed that selective suppression of the activity of neurons projecting from the mPFC to the vlPAG (projection neurons) abolished placebo analgesia.
MOR-positive neurons form direct synaptic connections with projection neurons and typically inhibit their activity. However, placebo administration relieves this inhibition. The disinhibited projection neurons were found to activate the descending pain inhibitory system, resulting in placebo analgesia.
Cui said, "I think detection of neuronal activity similar to human neuropsychiatric activity in the rodent brain was significant. Although previous placebo studies have been conducted mainly in humans, we used our original small-animal functional neuroimaging analysis method to demonstrate that the relevant brain regions in rodents were almost identical to those identified in human placebo studies, leading to detailed elucidation of the underlying molecular neurological mechanisms."
Journal Information
Publication: Science Advances
Title: Opioidergic activation of the descending pain inhibitory system underlies placebo analgesia
DOI: 10.1126/sciadv.adp8494
This article has been translated by JST with permission from The Science News Ltd. (https://sci-news.co.jp/). Unauthorized reproduction of the article and photographs is prohibited.




