This is the eighth installment in a series of stories in which Yutaka Minagawa, a young employee of a trading company, reads and analyzes proposal papers by the Center for Low Carbon Society Strategy (LCS). In the last installment, Minagawa looked at energy consumption and learned about the technological developments that support the rapidly progressing information society. In this installment, Minagawa interviews Researcher Mitsuo Koshi and Researcher Hiroshi Iwasaki about technologies for directly capturing carbon dioxide (CO2) from the atmosphere, which is considered essential for the realization of a decarbonized society by 2050.

Absorbed by reacting with a strongly alkaline solution — High temperatures of 900℃ are required for desorption
Minagawa: Thank you for your time today. Recently, I talked with Deputy Director‐General Taniguchi and LCS Researcher Otomo about advances that are being made with a technology called Direct Air Capture (DAC), which collects CO2 directly from the atmosphere. Today I'd like to hear about the latest trends in DAC. Technologies that do not increase atmospheric CO2 concentrations are the key to realizing a decarbonized society, aren't they?
Koshi: That's correct. Technologies for recovering and removing greenhouse gases are referred to as 'negative emissions,' and they are attracting quite a bit of attention. DAC is one such technology.
Minagawa: I know that there are methods to capture CO2 in the exhaust gases from power plants, but DAC captures CO2 from the atmosphere, which seems difficult.
Koshi: CO2 concentrations in the atmosphere are as low as 0.04%, so the DAC technologies are quite different from the technologies that capture high‐concentration CO2 of 10‐15% from emission sources. Since Professor Klaus Lackner of Arizona State University in the United States proposed DAC in 1999, there have been constant discussions about its effectiveness and feasibility.
Minagawa: How far has research and development progressed? Are overseas venture companies moving forward with DAC development?
Koshi: It has been calculated that one gigaton (1 billion tons) of CO2 must be collected annually by 2050 for the entire world to achieve decarbonization, but, currently, only about 10,000 tons of CO2 can be collected via DAC, which is nowhere near enough. Although DAC has yet to be put into practical use in Japan, it is a technology that will be essential in the future, so I think it is extremely significant that we are developing it.
Minagawa: Can you tell me how DAC captures CO2?
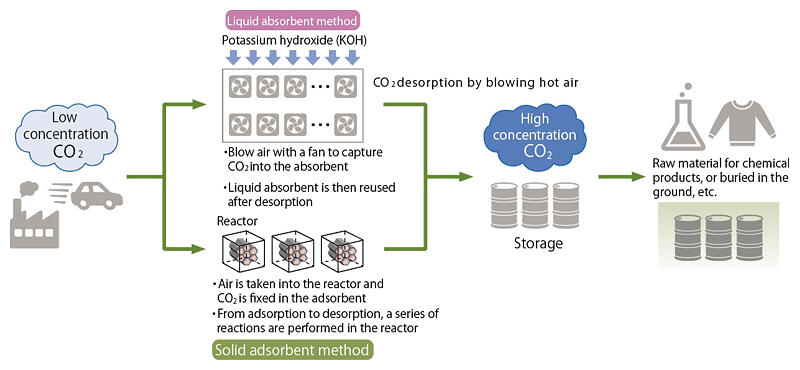
Koshi: Worldwide, there are two main technological methods that are being developed. One method uses an absorbent solution, the other a solid adsorbent. (Figure 1) In the method with the liquid absorbent, CO2 is absorbed in a strong alkaline solution and then reacted with calcium hydroxide to form calcium carbonate solids. When heat is applied to these solids, they become CO2 and calcium oxide, and the CO2 can be recovered.
Figure 1: Overview of DAC technologies
Minagawa: Technologies that absorb CO2 seem to be the key here.
Koshi: To efficiently incorporate CO2 into a liquid, a huge amount of surface area is required. In the method used by Carbon Engineering, a Canadian company, the surface of the air‐contact device, which is devised to improve gas‐liquid contact, will be wetted with potassium hydroxide, and then, air will be applied to it with a huge fan to make it react. In their plan, four fans with a diameter of 5 meters will be stacked vertically and then the fans will stretch horizontally for 2 kilometers.
Minagawa: Wow! So the structure would be 20 meters high, 2 kilometers wide, and filled with fans. The size of it is unimaginable.
Koshi: Even with this system, it would only capture a mere 900,000 tons of CO2 per year, which is less than the required amount. Another disadvantage is that high temperatures of 900℃ are required for the process of baking the solid calcium carbonate, and a huge amount of heat energy is used.
Adsorption on the surface of precisely porous materials provide challenges in the form of production costs and number of cycles
Minagawa: I see. So what about the method that uses a solid adsorbent?
Koshi: The adsorbent in this method immobilizes molecules on the surface of a porous material that is highly reactive with CO2. Climeworks, a Swiss company, uses an adsorbent with amine molecules fixed on the surface of nanofiber cellulose. The adsorbent is then placed in a 2.5m x 2.5m x 1.2m reactor, and a fan draws in air to fix the CO2 on the adsorbent. After this, the remaining air in the box is vented out, and the box is heated to 100℃ to desorb the CO2. A series of adsorption and desorption processes are possible with this reactor.
Minagawa: This method seems more promising because it requires less heat energy.
Iwasaki: However, in order to capture 900,000 tons of CO2 per year, approximately 11,000 reactors would be required for the adsorption and desorption operations.
Koshi: In addition, there is the disadvantage of the high cost of the adsorbent. Precise control of the adsorbent's surface structure is required to increase its surface area, which incurs a certain amount of cost. There is also the problem that the adsorbent deteriorates when heat is applied.
With Carbon Engineering's absorption liquid, it costs about ¥35 to capture 1 kg of CO2, while with Climework's adsorption method it costs about ¥117 to capture 1 kg of CO2, which is much higher. For DAC to be economically viable, it must withstand at least several tens of thousands of cycles.
Minagawa: Well... in that case it seems that there are still issues with both methods.
A new method that does not require heat energy — Expectations for research, but CO2 storage locations are also an issue
Iwasaki: Solid adsorbents have development potential for a variety of reasons, so there is intensive research on them around the world. And, as a new idea, it has also been said that we should consider methods that do not use thermal energy when desorbing CO2.
Minagawa: Is that possible?
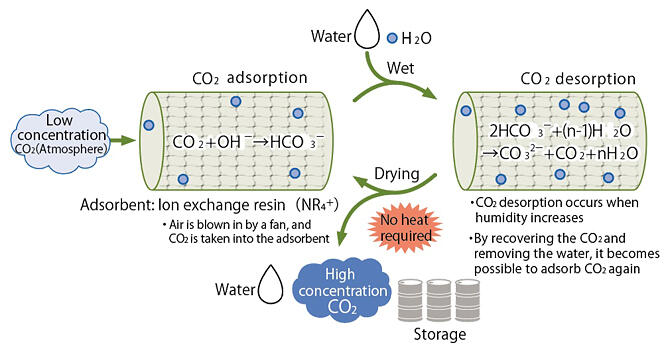
Iwasaki: There is a method called moisture swing adsorption (MSA) which is actively being studied in China and the United States (Figure 2).
This method uses a solid adsorbent, but utilizes the cohesive energy of hydrated water to desorb the CO2.
Figure 2: Moisture swing adsorption (MSA) mechanism
Minagawa: What do you mean?
Iwasaki: When water is added to the adsorbent, hydrated water condenses around the CO2‐fixing adsorbent, and the water's cohesive energy is used to desorb the CO2. Once it is dried, the adsorbent will once again adsorb CO2. Another major advantage of this method is that heat is not required and adsorbent degradation is minimized.
Minagawa: It sounds easy when I'm just listening to the explanation, but I'm sure it's actually quite difficult.
Koshi: It's possible in theory, but it isn't an established technology, so we are researching the technologies that will make it possible.
Iwasaki: Throughout the world, this is still laboratory‐scale research. The water problem is extremely important. The water is added as steam, and we must confirm via demonstrations whether we have to evaporate it and whether the sprayed water is evenly applied.
Minagawa: I have great expectations for the future technological development of these technologies. But, even if we successfully capture the CO2, what happens after that? I've heard that storing it underground is also being considered, but it doesn't seem like that will be realized.
Iwasaki: If we store it in Japan then the costs will be low, but if it is taken overseas then it will be seen as waste, so there will also be political issues such as cooperation with the relevant countries.
Koshi: CO2 is a global issue, so we have to think globally about an implementation system. In any case, DAC is the key to realizing a decarbonized society by 2050, so we think that it is important to move forward with the research right now in Japan so that we can practically apply it.
Minagawa: My company also needs to pay attention to new businesses and to think about investments and business alliances as soon as possible. Thank you for your time today.
(TEXT: Akane Iwasaki; PHOTO: Seiko Kusunoki, Tomokazu Fukui)






